Shoulder:Sepsis of the Shoulder
Contents
- 1 Bullet Points
- 2 Key Words
- 3 Introduction
- 4 Microbiology
- 5 Incidence and Prevalence
- 6 Risk Factors
- 7 Diagnosis
- 8 Clinical Presentation
- 9 Treatment
- 10 References
- 11 Sternoclavicular Joint Infection
- 12 Introduction
- 13 Etiology
- 14 Epidemiology
- 15 Pathophysiology
- 16 History and Physical
- 17 Evaluation
- 18 Treatment / Management
- 19 Differential Diagnosis
- 20 Prognosis
- 21 Complications
- 22 Deterrence and Patient Education
- 23 Enhancing Healthcare Team Outcomes
- 24 Questions
- 25 References
Bullet Points
- Periprosthetic shoulder infection is rare but potentially devastating. The rate of periprosthetic shoulder infection is increased in cases of revision procedures, reverse shoulder implants and comorbidities. One specific type of periprosthetic shoulder infection is the occurrence of low-grade infections caused by non-suppurative bacteria such as Cutibaterium acnes or Staphylococcus epidemermidis.
- Treatment success depends on microorganism identification, appropriate surgical procedures and antibiotic administration efficiency. Post-operative early periprosthetic shoulder infection can be treated with simple debridement, while chronic periprosthetic shoulder infection requires a one- or two-stage revision procedure. Indication for one-time exchange is based on pre-operative identification of a causative agent. Resection arthroplasty remains an option for low-demand patients or recalcitrant infection.
Key Words
Infection; Arthroplasty; Shoulder; Cutibacterium acnes; Propionibacterium; Revision.
Introduction
While more than 66 000 prosthetic shoulder procedures were performed in 2011 in the United States, the rate of post-operative infection seems to remain stable with 0.98% of cases.[2][3][4] However, when infection occurs, this complication is always devastating with significant clinical and socioeconomic consequences. [2]2 The rate is higher after revision surgery than after a primary procedure and reaches close to 5% in cases of reverse shoulder arthroplasty.[5][6] Patients undergoing primary reverse shoulder arthroplasty are found to have a six times greater risk of infection compared with patients having primary unconstrained total shoulder arthroplasty.[6] Arthroplasties for trauma are more at risk of infection than those from other causes.[6] Comorbidities such as coagulopathy, renal failure, diabetes, lupus erythematosus, rheumatoid arthritis, intra-articular steroid injections and corticosteroid therapy increase the risk of periprosthetic shoulder infection.[7] Periprosthetic shoulder infection is the major cause for revision within the first two post-operative years after an arthroplasty.[8]
Microbiology
Prevention
Antibiotic prophylaxis is not specific to shoulder arthroplasty compared with other arthroplasties. In many countries intravenous cephalosporine (2 g) administration is mandatory 30 minutes before the skin incision. However, some authors recommend a single 160 mg of gentamicin by intra-articular injection at the end of the procedure to reduce the risk of periprosthetic shoulder infection.[9] Saltzman et al. have shown that pre-operative preparation of the surgical site with chlorhexidine gluconate and 70% isopropyl alcohol was more effective than iodine povacrylex and 74% isopropyl alcohol and povidone-iodine to eliminate overall bacteria, and that the two first ones were more effective than povidone-iodine regarding coagulase-negative Staphylococcus[10].
Hair removal is commonly performed before orthopaedic procedures and the use of razors is classically discouraged because micro-abrasions are created by shaving. However, removal of axillary hairs for shoulder surgery did not prove to have any effect on the cell-count of Cutibacterium acnes before surgical preparation.[11]
Cutibacterium (Propionibacterium) acnes
Cutibacterium acnes (formerly Propionibacterium acnes) is a non-spore-forming, anaerobic, gram-positive bacillus. It is of low virulence and, therefore, can be a commensal in the lipid-rich sebaceous follicles and deep layers of the skin, conjunctiva, external auditory canal, respiratory tract and intestinal tract.[12][13] Cutibacterium acnes mostly colonises the pilosebaceous follicles in the skin of the upper-body, especially the head, neck, shoulders and axilla.[14][15] However, pathogenic activity of the organism has been shown in conditions such as meningitis, septic arthritis, osteomyelitis, chronic prostatitis and sarcoidosis. Cutibacterium acnes expresses proteins required for cell-adherence, which are also antigenic and therefore capable of initiating an inflammatory response of the host’s innate immune system within the joint.[12] Cutibacterium acnes also secretes cytotoxic chemicals and enzymes designed to degrade body tissues, which can be harmful within the shoulder capsule.[12] Furthermore, Cutibacterium acnes is also able to secrete and live within an extracellular polysaccharide biofilm aiding joint colonisation and micro-colony formation, as well as avoid phagocytosis and survive macrophage engulfment.[12] The combination of the bacterium’s upper-limb distribution and role in prosthetic joint infection is now forcing shoulder surgeons to consider Cutibacterium acnes as an ‘orthopaedic pathogen’.[13]
Drug resistance
Patient knowledge about Cutibacterium acnes is mostly limited to acne vulgaris, the skin infection present in the vast majority of young adults and teenagers.[12] Hormonal changes and genetics cause sebaceous gland inflammation, dysfunction and proliferation providing an environment for the opportunistic Cutibacterium acnes.[12] The treatment of this skin condition with experimental courses of broad-spectrum antibiotics, often combined with poor compliance, is thought to be a contributor to antibiotic resistance.[14] Gold standard antibiotics such as penicillins, vancomycin, tetracyclines and erythromycin often eradicate the bacterium, however, over half of Cutibacterium acnes cultures now carry resistance to more than one of the above antibiotics.[14]
Incidence and Prevalence
Over the past decade, Cutibacterium acnes has become increasingly recognised as a cause of infection in orthopaedic surgery, especially when prosthesis is involved.[16] With regards to shoulder replacement surgery, one study reported infection rates for primary shoulder replacement to be less than 4%, but following reverse replacement, to be as high as 18%. In this study the most common bacteria identified were Staphylococcus epidermis and Cutibacterium acnes.[17] However, in a retrospective review over 7 years carried out in Canada, 80 patients were identified who underwent joint cultures after primary shoulder arthoplasty, and Cutibacterium acnes was found to be the only significant infectious agent in 25% of participants, making it the most common pathogenic organism.[18] A study of periprosthetic joint infections after total shoulder arthroplasty in the last 33 years found that Staphylococcus was the dominant organism in the vast majority of cases, whereas from 2001 - 2008 the incidence of Cutibacterium acnes was found to be almost as high as Staphylococcus.[19] This increasing incidence could be the result of changes in the microbiology of shoulder infections, heightened awareness of the organism, better surveillance or improved laboratory diagnostic techniques[19] Cutibacterium acnes appears to be a prominent aggressor and is becoming more prevalent.
Risk Factors
Recorded risk factors for Cutibacterium acnes infection are male gender, surgery of the upper body (especially the shoulder), increased surgery duration and, interestingly, being the first surgery of the day.[4][20] The predominance of Cutibacterium acnes in shoulder infections has been linked with the presence of the axillary lymph nodes.[21] A study that involved culturing the epidermis of wounds during revision surgery found that 16 out of 18 involved males had Cutibacterium acnes infection, whereas only 7 out of 12 female subjects were tested positive.[22] Further studies have also found that male gender and prior prosthetic implants are significant risk factors for Cutibacterium acnes infection.[18][20] The male predisposition has been linked with the habitat of Cutibacterium acnes in the hair follicles and, therefore, the upper body of males would harbour more bacteria.[20] Furthermore, males were found to have over 2.5 times higher risk than females after shoulder arthroplasty.[23] Interestingly, if shoulder arthroplasty was performed following trauma rather than elective surgery, the risk of infection was nearly 3 times higher. Again, reverse shoulder arthroplasty was shown to increase the risk of infection with Cutibacterium acnes. Younger age was also identified as a risk factor, with each year of increasing age causing a risk reduction of 5%. Body Mass Index and diabetes mellitus, which are usually associated with increased risk of infection, were not identified as risk factors.[23] From the literature it is evident that patients at the highest risk of Cutibacterium acnes infection are young people, males, those who have undergone upper limb surgeries (especially reverse shoulder replacements) and those who have a surgery following trauma.
Diagnosis
Despite laboratory and investigative advances, the diagnosis of shoulder infections due to relatively low-virulence organisms, like Cutibacterium acnes, remains difficult. The non-specific clinical presentation, inadequate culture performance and the inability to accurately interpret positive cultures mean that diagnosis can be delayed.[18] Researchers have found that inflammatory markers usually indicative of infection may not be raised in Cutibacterium acnes infection.[24] In 2014, in a clinic in the US, C-reactive protein and erythrocyte sedimentation rate were both raised in only 10% of patients who were identified as having Cutibacterium acnes infections of the shoulder.[24] In a further study, the sensitivities of C-reactive protein and erythrocyte sedimentation rate in the shoulder were found to be 42% and 16% respectively, whereas in the lower limbs the sensitivities for C-reactive protein and erythrocyte sedimentation rate were 88% and 75% respectively.[25] Thus, although the sensitivity and specificity of these inflammatory markers are strong in the lower limbs, they are poor indicators of post-operative shoulder infections.[24] These data fit well with the distribution of Cutibacterium acnes and its indolent nature. It has been further suggested that due to its slow growth Cutibacterium acnes needs longer culture times (up to two weeks), making it harder to spot.[25] Furthermore, lowering the threshold for diagnosis (concentration of neutrophils in the tissues) would increase sensitivity because of the low inflammatory behavior of Cutibacterium acnes.[25]
Clinical Presentation
A common assumption is that the origin of Cutibacterium acnes infection is in the dermis of the patient’s skin and that it travels from the surface to contaminate the surgical site.[22] However, surface sterilisation before surgery does not eradicate the bacterium, as the organism actually resides deep in the sebaceous glands. Scalpel incisions slice through these follicles, allowing for seeding and leakage of the bacteria.[22] Innovative methods of sterilization may, therefore, be required to penetrate the dermis and reduce Cutibacterium acnes spread. Further, the need for intraoperative screening of wounds and prolonged cultures to assess the risk of developing a postoperative shoulder infection has been emphasised.[22] For low-grade Cutibacterium acnes shoulder infections pain may be the only symptom, followed by stiffness with all other signs of infection being absent.[26] Periprosthetic joint infections can be extremely serious and may result in loss of normal function of the joint or even progress to sepsis.[17]
A study, which focussed on postoperative pain, found that patients felt pain for an average of 3 months before diagnosis of Cutibacterium acnes infection and that the mean time from surgery to diagnosis was almost 2 years.[14] It was also suggested that the variance in the clinical presentation of shoulder infections may not only be due to the non-specific clinical manifestation of Cutibacterium acnes, but also due to co-infection with other bacteria within the surgical site.[14]
Treatment
Debridement
Debridement, irrigation and multiple deep samples may be proposed in cases of acute infection in order to save the prosthesis.[27] Treated eight cases of acute infection with debridement and succeeded when it was performed within eight days after the diagnosis. Authors concluded that the earlier the debridement is done, the more effective it is in eradicating the infection. This procedure can be repeated based on the patient’s response.[28] Moreover, mobile parts of the prosthesis may be exchanged during the procedure, especially in case of reverse shoulder arthroplasty (glenosphere, polyethylene liner) providing better access for debridement. Then, an appropriate antibiotics regime is required for a minimum of four weeks.[28][29][30] However, the success rate reported in the literature is only in the range from 50% to 95%.[28][29][31]
Cement spacer
An antibiotic-loaded cement spacer can be used either permanently or as the first step of a two-stage revision procedure. In this case, it maintains the space and soft-tissue tension for re-implantation and theoretically releases antibiotics to decrease the growth of microorganisms. Antibiotic mean concentration peak is reached at day 1 and dramatically decreases during the following seven days.[32] Levy et al. described a ‘functional cement spacer’ model, which is made of a hemi-arthroplasty coated with cement.[33] In their series of fourteen patients initially chosen for a two-stage procedure, nine did not undergo a prosthesis re-implantation because of satisfactory clinical outcomes. On the other hand, Verhelst et al. did not prove any difference between patients with a cement spacer and resection arthroplasty regarding infection control and clinical outcomes[34]. Complications of the cement-spacer such as breakage, glenoid erosion or dislocation have been reported.[34]
One-stage revision arthroplasty
Based on the experience of knee and hip infection management, a single-stage exchange is proposed as a reasonable option when the infectious microorganism is satisfactorily identified. The advantages are a reduced hospital stay, costs, period of antibiotic administration and the best clinical outcomes (Table).[10][17][27][31][30][35][36][37][38] Klatte et al.33 [17]reported the outcomes of the largest single-centre series of 35 patients with a mean follow-up of 4.7 years. The authors excised infected tissues, thoroughly irrigated the wound using pulsatile lavage with polyhexanide before re-implantation and delivered specific intravenous antibiotherapy for an average of two weeks post-operatively. The success rate was more than 90%. No recurrence was observed in the series of Coste, Ince and Cuff.[27][35][36] The presence of a productive fistula seems not to be a contraindication for many authors.[17][37] Beekman et al. performed a one-stage revision in 11 cases of reverse shoulder arthroplasties, among which eight had a fistula, and achieved a success rate of 90%.[37]
Two-stage revision arthroplasty
In a medically stable patient with a high demand, a two stage revision procedure is generally accepted (Table).[17][27][31][30][39][40][41] It is highly recommended when the microorganism responsible for the infection is unknown. The first step consists of infection eradication after prosthetic removal: an antibiotic-loaded cement spacer is often implanted and general antibiotics are administrated, secondarily adapted to the micro-organism(s) identified. Antibiotics are generally continued for six to eight weeks. Markers such as C-reactive protein or interleukin-6 have been shown to be valuable to predict the eradication of infection and, so, the time of re-implantation.[42][43] However, interleukin-6 seems to be normalized faster than C-reactive protein and allows earlier revision for better outcomes.[42] An iterative irrigation and debridement could be proposed in case of persistent infection. For re-implantation reverse shoulder arthroplasty has been gaining ground in recent years as the implant of choice. First, it allows a larger debridement at the first stage with less concern for soft-tissue preservation. Secondly, it offers the possibility of addressing the glenoid bone defect with or without bone graft. Shirwaiker et al. reported that there is still uncertainty whether two-stage revision is superior to one-stage (Figure 1)[44].
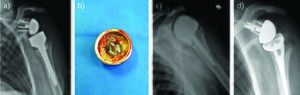
Resection arthroplasty
Shoulder resection should remain a salvage procedure for frail or low-demand patients and recalcitrant infection. It offers the option of a single definitive procedure for infection eradication (Figure 2). It has been shown that functional results are poor, but pain relief is achieved in more than 50% of cases.[10][45] Rispoli et al. reported a mean active elevation of 70 degrees at long-term follow-up after anatomical shoulder arthroplasty removal.[45] Verhelst et al. demonstrated that preservation of the tuberosities is a predictive factor for better results, because it can avoid antero-superior subluxation of the humerus.[2] In cases of reverse shoulder arthroplasty, Jacquot et al. did not improve functional outcomes after removal of the implant and identified a high rate of post-operative complications.[29] Bone loss and soft-tissue impairment after such constrained prostheses could partly explain these findings. Despite Jacquot[29] and Coste’s[27] studies, the literature reports a high rate of infection eradication reaching more than 90% of cases.[2][10][27][30] [46][47]
References
- ↑ 1.0 1.1 1.2 Bonnevialle N, Dauzères F, Toulemonde J, Elia F, Laffosse J-M, Mansat, P. Periprosthetic shoulder infection: an overview EFORT Open Rev. 2017;2:104–9
- ↑ 2.0 2.1 2.2 2.3 Schairer WW, Nwachukwu BU, Lyman S, Craig EV, Gulotta LV. National utilization of reverse total shoulder arthroplasty in the United States. J Shoulder Elbow Surg 2015;24:91-7
- ↑ Padegimas EM, Maltenfort M, Ramsey ML, Williams GR, Parvizi J, Namdari S. Periprosthetic shoulder infection in the United States: incidence and economic burden. J Shoulder Elbow Surg 2015;24:741-6
- ↑ 4.0 4.1 Day JS, Lau E, Ong KL, Williams GR, Ramsey ML, Kurtz SM. Prevalence and projections of total shoulder and elbow arthroplasty in the United States to 2015. J Shoulder Elbow Surg 2010;19:1115-20
- ↑ Zumstein MA, Pinedo M, Old J, Boileau P. Problems, complications, reoperations, and revisions in reverse total shoulder arthroplasty: a systematic review. J Shoulder Elbow Surg 2011;20:146-57
- ↑ 6.0 6.1 6.2 Morris BJ, O’Connor DP, Torres D, et al. Risk factors for periprosthetic infection after reverse shoulder arthroplasty. J Shoulder Elbow Surg 2015;24:161-6
- ↑ Smucny M, Menendez ME, Ring D, Feeley BT, Zhang AL. Inpatient surgical site infection after shoulder arthroplasty. J Shoulder Elbow Surg 2015;24:747-53
- ↑ Portillo ME, Salvadó M, Alier A, Sorli L, Martínez S, Horcajada JP, Puig L. Prosthesis failure within 2 years of implantation is highly predictive of infection. Clin Orthop Relat Res 2013;471:3672-8
- ↑ Lovallo J, Helming J, Jafari SM, Owusu-Forfie A, Donovan S, Minnock C, Adib F. Intraoperative intra-articular injection of gentamicin: will it decrease the risk of infection in total shoulder arthroplasty? J Shoulder Elbow Surg 2014;23:1272-6
- ↑ 10.0 10.1 10.2 10.3 Saltzman MD, Nuber GW, Gryzlo SM, Marecek GS, Koh JL. Efficacy of surgical preparation solutions in shoulder surgery. J Bone Joint Surg Am. 2009;91:1949-53
- ↑ Marecek GS, Weatherford BM, Fuller EB, Saltzman MD. The effect of axillary hair on surgical antisepsis around the shoulder. J Shoulder Elbow Surg 2015;24:804-8
- ↑ 12.0 12.1 12.2 12.3 12.4 12.5 Aubin GG, Portillo ME, Trampuz A, Corvec S. Propionibacterium acnes, an emerging pathogen: From acne to implant-infections, from phylotype to resistance. Med Mal Infect. 2014;44:241-50
- ↑ 13.0 13.1 Levy O, Iyer S, Atoun E, Peter N, Hous N, Cash D, Musa F, Narvani AA. Propionibacterium acnes: an underestimated etiology in the pathogenesis of osteoarthritis? J Shoulder Elbow Surg. 2013;22:505-11
- ↑ 14.0 14.1 14.2 14.3 14.4 Millett PJ, Yen YM, Price CS, Horan MP, van der Meijden OA, Elser F. Propionibacterium acnes infection as an occult cause of postoperative shoulder pain: a case series. Clin Orthop Relat Res. 2011;469:2824-30
- ↑ Lorillou M, Martha B, Chidiac C, Ferry T, Lyon B, Joint Infection Study Group. Chronic Propionibacteriom acnes prosthesis joint infection manifesting as a large abscess with gas, without prosthesis loosening. BMJ Case Rep 2013; 2013:bcr2013201090
- ↑ Crane JK, Hohman DW, Nodzo SR, Duquin TR. Antimicrobial susceptibility of Propionibacterium acnes isolates from shoulder surgery. Antimicrob Agents Chemother 2013;57:3424-6
- ↑ 17.0 17.1 17.2 17.3 17.4 17.5 Klatte TO, Junghans K, Al-Khateeb H, Rueger JM, Gehrke T, Kendoff D, Neumann J. Single-stage revision for peri-prosthetic shoulder infection: outcomes and results. Bone Joint J. 2013;95-B:391-5
- ↑ 18.0 18.1 18.2 Wang B, Toye B, Desjardins M, Lapner P, Lee C. A 7-year retrospective review from 2005 to 2011 of Propionibacterium acnes shoulder infections in Ottawa, Ontario, Canada. Diagn Microbiol Infect Dis. 2013 Feb;75:195-9
- ↑ 19.0 19.1 Singh JA, Sperling JW, Schleck C, Harmsen WS, Cofield RH. Periprosthetic infections after total shoulder arthroplasty: a 33-year perspective.J Shoulder Elbow Surg. 2012;21:1534-41
- ↑ 20.0 20.1 20.2 Patel A, Calfee RP, Plante M, Fischer SA, Green A. Propionibacterium acnes colonization of the human shoulder. J Shoulder Elbow Surg. 2009;18:897-902
- ↑ Achermann Y, Sahin F, Schwyzer HK, Kolling C, Wust J, Vogt M. Characteristics and outcome of 16 periprosthetic shoulder joint infections. Infection. 2013;41:613-20
- ↑ 22.0 22.1 22.2 22.3 Matsen FA, 3rd, Butler-Wu S, Carofino BC, Jette JL, Bertelsen A, Bumgarner R. Origin of propionibacterium in surgical wounds and evidence-based approach for culturing propionibacterium from surgical sites. J Bone Joint Surg Am. 2013;95:e1811-7
- ↑ 23.0 23.1 Richards J, Inacio MC, Beckett M, Navarro RA, Singh A, Dillon MT,Sodl JF, Yian EH. Patient and Procedure-specific Risk Factors for Deep Infection After Primary Shoulder Arthroplasty. Clin Orthop Relat Res. 2014;472:2809-15
- ↑ 24.0 24.1 24.2 Mook WR, Garrigues GE. Diagnosis and Management of Periprosthetic Shoulder Infections. J Bone Joint Surg Am. 2014 4;96:956-65
- ↑ 25.0 25.1 25.2 Grosso MJ, Frangiamore SJ, Ricchetti ET, Bauer TW, Iannotti JP. Sensitivity of frozen section histology for identifying Propionibacterium acnes infections in revision shoulder arthroplasty. J Bone Joint Surg Am. 2014;96:442-7
- ↑ Schneeberger AG, Yian E, Steens W. Injection-induced low-grade infection of the shoulder joint: preliminary results. Arch Orthop Trauma Surg. 2012;132:1387-92
- ↑ 27.0 27.1 27.2 27.3 27.4 27.5 Coste JS, Reig S, Trojani C, Berg M, Walch G, Boileau P. The management of infection in arthroplasty of the shoulder. J Bone Joint Surg Br. 2004;86:65-9
- ↑ 28.0 28.1 28.2 Duncan SF, Sperling JW. Treatment of primary isolated shoulder sepsis in the adult patient. Clin Orthop Relat Res 2008;466:1392-96
- ↑ 29.0 29.1 29.2 29.3 Jacquot A, Sirveaux F, Roche O, Favard L, Clavert P, Molé D. Surgical management of the infected reversed shoulder arthroplasty: a French multicenter study of reoperation in 32 patients. J Shoulder Elbow Surg 2015;24:1713-22
- ↑ 30.0 30.1 30.2 30.3 Romanò CL, Borens O, Monti L, Meani E, Stuyck J. What treatment for periprosthetic shoulder infection? Results from a multicentre retrospective series. Int Orthop 2012;36:1011-17
- ↑ 31.0 31.1 31.2 Sperling JW, Kozak TK, Hanssen AD, Cofield RH. Infection after shoulder arthroplasty. Clin Orthop Relat Res 2001;382:206-16
- ↑ Anagnostakos K, Wilmes P, Schmitt E, Kelm J. Elution of gentamicin and vancomycin from polymethylmethacrylate beads and hip spacers in vivo. Acta Orthop 2009;80:193-7
- ↑ Levy JC, Triplet J, Everding N. Use of a functional antibiotic spacer in treating infected shoulder arthroplasty. Orthopedics 2015;38:e512-e51
- ↑ 34.0 34.1 Verhelst L, Stuyck J, Bellemans J, Debeer P. Resection arthroplasty of the shoulder as a salvage procedure for deep shoulder infection: does the use of a cement spacer improve outcome? J Shoulder Elbow Surg 2011;20:1224-33
- ↑ 35.0 35.1 Ince A, Seemann K, Frommelt L, Katzer A, Loehr JF. One-stage exchange shoulder arthroplasty for peri-prosthetic infection. J Bone Joint Surg Br. 2005;87:814-8
- ↑ 36.0 36.1 Cuff DJ, Virani NA, Levy J, Frankle MA, Derasari A, Hines B, Pupello DR, Cancio M, Mighell M. The treatment of deep shoulder infection and glenohumeral instability with debridement, reverse shoulder arthroplasty and postoperative antibiotics. J Bone Joint Surg Br. 2008;90:336-42
- ↑ 37.0 37.1 37.2 Beekman PD, Katusic D, Berghs BM, Karelse A, De Wilde L. One-stage revision for patients with a chronically infected reverse total shoulder replacement. J Bone Joint Surg Br. 2010;92:817-22
- ↑ Grosso MJ, Sabesan VJ, Ho JC, Ricchetti ET, Iannotti JP. Reinfection rates after 1-stage revision shoulder arthroplasty for patients with unexpected positive intraoperative cultures.J Shoulder Elbow Surg. 2012;21:754-8
- ↑ Strickland JP, Sperling JW, Cofield RH. The results of two-stage reimplantation for infected shoulder replacement. J Bone Joint Surg Br. 2008;90:460-5
- ↑ Ortmaier R, Resch H, Hitzl W, Mayer M, Stundner O, Tauber M. Treatment strategies for infection after reverse shoulder arthroplasty. Eur J Orthop Surg Traumatol 2014;24:723-31
- ↑ Sabesan VJ, Ho JC, Kovacevic D, Iannotti JP. Two-stage reimplantation for treating prosthetic shoulder infections. Clin Orthop Relat Res 2011;469:2538-43
- ↑ 42.0 42.1 Coffey MJ, Ely EE, Crosby LA. Treatment of glenohumeral sepsis with a commercially produced antibiotic-impregnated cement spacer. J Shoulder Elbow Surg. 2010;19:868-73
- ↑ Villacis D, Merriman JA, Yalamanchili R, Omid R, Itamura J, Rick Hatch 3rd GF. Serum interleukin-6 as a marker of periprosthetic shoulder infection. J Bone Joint Surg Am. 2014;96:41-5
- ↑ Shirwaiker RA, Springer BD, Spangehl MJ, Garrigues GE, Lowenberg DW, Garras DN, Yoo JU, Pottinger PS. A clinical perspective on musculoskeletal infection treatment strategies and challenges. J Am Acad Orthop Surg. 2015;23:S44-S54
- ↑ 45.0 45.1 Rispoli DM, Sperling JW, Athwal GS, Schleck CD, Cofield RH. Pain relief and functional results after resection arthroplasty of the shoulder.J Bone Joint Surg Br. 2007;89:1184-7
- ↑ Weber P, Utzschneider S, Sadoghi P, Andress HJ, Jansson V, Müller PE. Management of the infected shoulder prosthesis: a retrospective analysis and review of the literature. Int Orthop 2011;35:365-73
- ↑ Atkins BL, Athanasou N, Deeks JJ, Crook DW, Simpson H, Peto TE, McLardy-Smith P, Berendt AR. Prospective evaluation of criteria for microbiological diagnosis of prosthetic-joint infection at revision arthroplasty. The OSIRIS Collaborative Study Group. J Clin Microbiol. 1998;36:2932-9
Sternoclavicular Joint Infection
Cover of StatPearls StatPearls [Internet].
Show details Search term Sternoclavicular Joint Infection David C. Tapscott; Michael D. Benham.
Author Information Last Update: June 2, 2020.
Introduction
The sternoclavicular joint is a saddle-shaped diarthrodial joint that joins the upper extremity appendicular skeleton to the axial skeleton. The large medial clavicle articulates with the superomedial manubrium and costal cartilage of the first rib, forming a joint with very little bony stability.[1][1] Within the joint is an intra-articular disc ligament composed of dense fibrous cartilage that provides structural support and prevents medial displacement of the clavicle.[2][2] The surrounding robust costoclavicular ligament and capsule offers an added layer of support.[2][2] The primary restraints to anterior and posterior translation of the joint are the anterior and posterior sternoclavicular ligaments.[3][3] Despite these restrictions, the sternoclavicular joint is actually very mobile and moves more than 30 degrees in the axial and coronal planes while having more than 45 degrees of rotation.[4][4] Functionally, it is quite similar to other amphiarthroses, such as the sacroiliac joint or pubic symphysis.[5][5]
Blood supply to the joint comes from the articular branches of the suprascapular and internal thoracic arteries.[1][1] The nerve to the subclavius muscle and the medial suprascapular nerves provide innervation to the sternoclavicular joint.[1][1]
Septic arthritis of the sternoclavicular joint is rare and represents less than 1% of all bone and joint infections.[5][6][5][6] In the majority of cases a sternoclavicular joint infection is associated with other systemic illnesses and/or general poor health status. Common concurrent issues include diabetes, intravenous drug use, immunosuppression, and rheumatoid arthritis.[7][7] While rare, prompt diagnosis and treatment are essential to prevent spread into the posteriorly located great vessels, mediastinum, and pleural space.[7][7]
Etiology
Ross et al. reviewed 180 cases of sternoclavicular joint septic arthritis and found that the most common offending bacteria were Staphylococcus aureus (49%), followed by Pseudomonas aeruginosa (10%) and Brucella melitensis (7%)[5].[5] In a similarly designed study, Brancos et al. reported sternoclavicular joint infections in a population of heroin addicts. They confirmed S. aureus and P. aeruginosa as the most common isolates.[8][8] In 2015, Jain et al. described tuberculosis (TB) as an unusual cause of sternoclavicular septic arthritis. They reported a small series of 9 men and 4 women, who eventually received a diagnosis of sternoclavicular septic arthritis secondary to TB.[9][9]
Epidemiology
In the general population, sternoclavicular septic arthritis accounts for less than 1% of bone and joint infections.[6][6] Interestingly, it accounts for 17% of septic arthritis cases amongst intravenous drug users.[5][5] The classic involved patient demographic consists of males in the fourth to fifth decade of life. In the previously mentioned case series by Ross et al., 73% of sternoclavicular joint infections involved males with a mean age of 45 years .[5][5] Interestingly, no risk factor for infection was identifiable in almost a quarter of the patients in the study.[5][5] Von Glinski et al. reported 13 cases of sternoclavicular joint infection involving eight men with a mean age of 37 years.[10][10]
Pathophysiology
Infection of the sternoclavicular joint can occur via direct inoculation of the joint or contiguous spread from a nearby area; however, the most common pathway is hematogenous spread via the blood-stream. This particular mechanism explains why the prevalence of this rare condition is so much more commonly seen in patients with a history of intravenous drug abuse.[11][11]
A healthy patient may be able to contract a spontaneous sternoclavicular joint infection. Sanelli et al. describe a case os staphylococcal septic arthritis that is associated with no known predisposing risk factor. That patient was able to be managed with medical therapy alone.[12][12]
There have been reports of patients contracting sternoclavicular joint infections in association with dialysis. Renoult et al. first reported on the association of hemodialysis and sternoclavicular joint infection. They reported two patients who were able to be treated non-operatively after addressing the condition.[13][13] Renal failure may further immunosuppress and predispose such patients to a sternoclavicular joint infection.
History and Physical
An extensive history involving the assessment of systemic symptoms is vital, as many of the conditions that affect the sternoclavicular joint are systemic. The clinician should question the patient regarding a family history of sternoclavicular arthritis, intravenous drug use, and systemic complaints such as subjective fevers, chills, night sweats, and malaise. Pain localized to the sternoclavicular joint or medial clavicle is a high-risk patient, or an individual with systemic symptoms should raise suspicion for infection.
On physical examination, an infected sternoclavicular joint is likely to be swollen, warm, tender, and erythematous. Patients are painful both with direct palpation of the joint as well as passive/active range of motion of the ipsilateral shoulder. Typically, sternoclavicular joint septic arthritis is a unilateral condition, while inflammatory arthritis such as rheumatoid arthritis affects both joints simultaneously. One should pay attention to any fluctuance, joint translation, joint asymmetry, and bony enlargement. Joint asymmetry should raise concern for a sternoclavicular joint dislocation, which may represent a surgical emergency, if directed posteriorly with neurovascular changes.[14][14]
Evaluation
As in most cases of orthopedic infections, an elevated white blood cell count (WBC), erythrocyte sedimentation rate (ESR), and C-reactive protein (CRP) present as elevated. These markers are relatively nonspecific and may increase in cases of gout, systemic illness, or infection of another joint. As previously stated, these patients may be immunosuppressed and may be unable to elicit a systemic response that produces impressive inflammatory labs. It is also reasonable to routinely obtain blood cultures on these patients at the time of presentation as estimates are that nearly two-thirds of patients with sternoclavicular joint infections are systemically septic.[5][5]
Unfortunately, imaging studies for these patients are nonspecific and most helpful in ruling out other painful etiologies. If deemed necessary, an MRI is an excellent tool to evaluate for joint effusion, nearby abscess, or early osteomyelitis.[5] Radiographs of the sternoclavicular joint may be helpful, but likely only in the setting of osteomyelitis, as isolated septic arthritis would lack bony involvement by definition. Von Glinski et al. obtained chest radiographs in all patients in their series of 13 to assess surrounding tissues.[10][10]
CT scan may hold some additional value, as it may show earlier bony changes as well as demonstrate the presence of any peri-articular or retrosternal abscess. Von Glinski et al. described a series of 13 patients, of whom 10 had a periarticular abscess that was identifiable on a preoperative CT scan.[10][10]
The gold standard for diagnosing sternoclavicular joint septic arthritis remains arthrocentesis. Aspirated joint fluid should undergo evaluation for crystals, cell count, percentage of neutrophils, and the presence of bacteria using gram stain and culture. A total nucleated cell count greater than 50000 with over 90% neutrophils with no crystals is concerning for an infection. A positive gram stain or culture is virtually diagnostic regardless of the other synovial fluid analyses.
Rochuae et al. reported on a group of sternoclavicular joint arthritis patients and found that only 53% had proven bacteremia at the time of evaluation.[15][15] Blood cultures are a reasonable adjunct to obtain during patient evaluation. However, this relatively low positivity rate means blood cultures cannot be solely relied upon to isolate the offending organism.
Treatment / Management
While there are reports of successful treatment with needle arthrocentesis and antibiotics, the mainstay of treatment for sternoclavicular joint infections is surgical irrigation and debridement with tailored intravenous antibiotic therapy.[9][16][9][16] Care should be taken to evaluate the surrounding chest wall for loculated abscesses that may be a source of continued infection at the time of operative irrigation and debridement. Particular aggressive organisms or recalcitrant infections may require resection of the medial clavicle with possible soft tissue coverage given the subcutaneous nature of the sternoclavicular joint. When resecting the medial clavicle, the vital retrosternal structures, as well as the surrounding ligamentous structures, must be protected.[1][1] If the supporting ligamentous structures are damaged, pain and instability can result.[17][17]
In a series of 10 patients, who underwent resection arthroplasty for sternoclavicular joint septic arthritis, Chun et al. reported good functional results with intramedullary ligament reconstruction and 4 to 8 weeks of intravenous antibiotics.[18][18] There are multiple studies available in the literature detailing joint resection techniques as well as closure methods.[9][19][18][9][19][18] Whitlark et al. describe a single-stage washout with implantation of antibiotic laden beads and a good outcome in a single case.[20][20]
Less invasive management may be an option in less advanced disease or an immunocompetent host. Multiple authors have reported cases or case series that were manageable with antibiotics alone.[12][13][13][12] However, due to a lack of larger studies with longer follow-ups, the success rate of this treatment technique is not well described. Isolated aspiration or needle lavage has also been suggested, but lacks significant literature to prove its reliability.
If there becomes a significant soft tissue defect from adequate debridement, additional flap coverage may be required. Although uncommon, it is possible to need a soft tissue transfer. Opoku-Agyeman et al. reported on several surgical configurations for pectoralis major flaps in the reconstruction of sternoclavicular defects.[21][21] These techniques include a pectoralis split and advance, split and rotate, or entire pectoralis harvest and transfer. These techniques may be considered in surgical planning, if significant debridement is required. Primary muscle flap closure has demonstrated excellent outcomes in a series of 40 patients, as described by Kachala et al.[19][19]
A multidisciplinary approach, including the involvement of an infectious disease, may optimize the patient outcome. Postoperatively, or in the case of medical management, the mainstays of antibiotic therapy for any septic arthritis should be a post-operative course of antibiotics of at least two weeks depending on the offending agent and the recommendations of your infectious disease team.[22][22]
Differential Diagnosis
The differential diagnosis of sternoclavicular joint infection includes sternoclavicular joint arthritis, clavicular osteomyelitis, clavicular fracture, sternal fracture, pleuritis, gout, dislocation, condensing osteitis of the clavicle, mediastinitis, or rib fracture.
Prognosis
The prognosis for patients with sternoclavicular joint infections recognized promptly and treated appropriately is excellent. Burkhart et al. reported a case series of sternoclavicular joint infections treated with joint resection with no resulting limitations of the affected upper extremity.[23][23] Untreated infections can develop into extrapleural or intrathoracic abscesses and quickly become life-threatening, if the retrosternal structures are involved.[1][1]
Initial post-operative care will revolve around clearing the infection and obtaining adequate soft tissue healing. After achieving a source control, the function of the limb is the primary focus. In an excellent review of sternoclavicular joint instability, Sewell et al. describe physical therapy as being beneficial for patients with SC joint instability.[24][24] Being able to offer treatment as a postoperative adjunct for a patient experiencing slow recovery may increase patient rehabilitation and improve the overall outcome.
Complications
Complications include: the need to repeat a surgery, osteomyelitis, skin loss over the affected area requiring flap coverage, the joint instability, need for joint reconstruction, inability to clear the infection, and progression to the chest wall or mediastinal involvement. Patients may have chronic pain with shoulder range of motion or pain with heavy lifting in cases of chronic infections or delayed diagnoses.
Deterrence and Patient Education
Correcting modifiable risk factors and providing education regarding the dangers of intravenous drug use may lower the incidence of sternoclavicular joint infections.
Enhancing Healthcare Team Outcomes
The diagnosis and management of patients with sternoclavicular joint infection are complex and best done with an interprofessional team. The nurses may be the first to identify the pathology in in-patients, especially in those with central catheters. Outpatients usually present to the emergency room.
A combination of history, physical examination, and laboratory findings are necessary to make the diagnosis of sternoclavicular joint infection. Many of these patients likely present to primary care providers or emergency departments, so a basic knowledge of this condition is required to make a prompt diagnosis and avoid the spread of the infection or a poor outcome. The services of radiology are vital, as imaging is often used to confirm the diagnosis. The infectious disease expert should be consulted for antibiotics and the duration of therapy. A surgeon is usually involved, as many patients require drainage and debridement. The board-certified infectious disease pharmacist should pay attention to culture results and ensure that the patient is on the appropriate antibiotics compared to antibiogram data. The pharmacist can also verify dosing and duration, and check for potential drug interaction, informing the team, if there are any issues. Nursing can assist the clinician during the case, verifying patient compliance, acting as a bridge to the clinician for the patient and other providers (e.g., physical therapist), and providing patient counsel.
Prompt referral to an orthopedic or thoracic surgeon is vital, if the infection has resulted in osteomyelitis of the sternum or clavicle. Orthopedic and thoracic surgeons must provide education on the ramifications of a missed diagnosis to other healthcare providers. Close communication between the team members is vital for improving outcomes. [Level 5]
Questions
To access free multiple choice questions on this topic, click here.
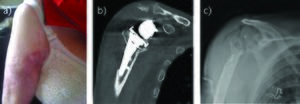
Sternoclavicular infection Figure
Sternoclavicular infection. Image courtesy S Bhimji MD Sternoclavicular joint anatomy Figure
Sternoclavicular joint anatomy. Image courtesy S Bhimji MD Sternoclavicular joint infection Figure
Sternoclavicular joint infection. Image courtesy S Bhimji MD Go to:
References
1. Higginbotham TO, Kuhn JE. Atraumatic disorders of the sternoclavicular joint. J Am Acad Orthop Surg. 2005 Mar-Apr;13(2):138-45. [PubMed]
2. Logan C, Shahien A, Altintas B, Millett PJ. Rehabilitation Following Sternoclavicular Joint Reconstruction for Persistent Instability. Int J Sports Phys Ther. 2018 Aug;13(4):752-762. [PMC free article] [PubMed]
3. Spencer EE, Kuhn JE, Huston LJ, Carpenter JE, Hughes RE. Ligamentous restraints to anterior and posterior translation of the sternoclavicular joint. J Shoulder Elbow Surg. 2002 Jan-Feb;11(1):43-7. [PubMed]
4. Giphart JE, Brunkhorst JP, Horn NH, Shelburne KB, Torry MR, Millett PJ. Effect of plane of arm elevation on glenohumeral kinematics: a normative biplane fluoroscopy study. J Bone Joint Surg Am. 2013 Feb 06;95(3):238-45. [PubMed]
5. Ross JJ, Shamsuddin H. Sternoclavicular septic arthritis: review of 180 cases. Medicine (Baltimore). 2004 May;83(3):139-48. [PubMed]
6. Bar-Natan M, Salai M, Sidi Y, Gur H. Sternoclavicular infectious arthritis in previously healthy adults. Semin. Arthritis Rheum. 2002 Dec;32(3):189-95. [PubMed]
7. Nusselt T, Klinger HM, Freche S, Schultz W, Baums MH. Surgical management of sternoclavicular septic arthritis. Arch Orthop Trauma Surg. 2011 Mar;131(3):319-23. [PMC free article] [PubMed]
8. Brancós MA, Peris P, Miró JM, Monegal A, Gatell JM, Mallolas J, Mensa J, García S, Muñoz-Gomez J. Septic arthritis in heroin addicts. Semin. Arthritis Rheum. 1991 Oct;21(2):81-7. [PubMed]
9. Jain A, Jajodia N, Aggarwal A, Singh J, Gupta S. Tuberculosis of the sternoclavicular joint. J Orthop Surg (Hong Kong). 2015 Dec;23(3):315-8. [PubMed]
10. von Glinski A, Yilmaz E, Rausch V, Koenigshausen M, Schildhauer TA, Seybold D, Geßmann J. Surgical management of sternoclavicular joint septic arthritis. J Clin Orthop Trauma. 2019 Mar-Apr;10(2):406-413. [PMC free article] [PubMed]
11. Jang YR, Kim T, Kim MC, Sup Sung H, Kim MN, Kim MJ, Kim SH, Lee SO, Choi SH, Woo JH, Kim YS, Chong YP. Sternoclavicular septic arthritis caused by Staphylococcus aureus: excellent results from medical treatment and limited surgery. Infect Dis (Lond). 2019 Sep;51(9):694-700. [PubMed]
12. Zanelli G, Sansoni S, Migliorini L, Donati E, Cellesi C. Sternoclavicular joint infection in an adult without predisposing risk factors. Infez Med. 2003 Jun;11(2):105-7. [PubMed]
13. Renoult E, Lataste A, Jonon B, Testevuide P, Kessler M. Sternoclavicular joint infection in hemodialysis patients. Nephron. 1990;56(2):212-3. [PubMed]
14. Morell DJ, Thyagarajan DS. Sternoclavicular joint dislocation and its management: A review of the literature. World J Orthop. 2016 Apr 18;7(4):244-50. [PMC free article] [PubMed]
15. Rodchuae M, Ruangpin C, Katchamart W. Clinical manifestations, treatment outcomes, and risk factors for sternoclavicular septic arthritis. Rheumatol. Int. 2017 May;37(5):819-824. [PubMed]
16. Szentkereszty Z, Pósán J, Petö K, Sápy P, Boros M, Takács I, Sz Kiss S. [Surgical management of sternoclavicular joint infections]. Magy Seb. 2007 Jan;60(1):514-7. [PubMed]
17. Rockwood CA, Groh GI, Wirth MA, Grassi FA. Resection arthroplasty of the sternoclavicular joint. J Bone Joint Surg Am. 1997 Mar;79(3):387-93. [PubMed]
18. Chun JM, Kim JS, Jung HJ, Park JB, Song JS, Park SS, Lee HS, Ahn TS. Resection arthroplasty for septic arthritis of the sternoclavicular joint. J Shoulder Elbow Surg. 2012 Mar;21(3):361-6. [PubMed]
19. Kachala SS, D'Souza DM, Teixeira-Johnson L, Murthy SC, Raja S, Blackstone EH, Raymond DP. Surgical Management of Sternoclavicular Joint Infections. Ann. Thorac. Surg. 2016 Jun;101(6):2155-60. [PubMed]
20. Whitlark JD, Kirollos JA, Jackson SM. Simplified Method for Treating Osteomyelitis of the Sternoclavicular Joint. Ann. Thorac. Surg. 2016 Mar;101(3):1211-2. [PubMed]
21. Opoku-Agyeman J, Matera D, Simone J. Surgical configurations of the pectoralis major flap for reconstruction of sternoclavicular defects: a systematic review and new classification of described techniques. BMC Surg. 2019 Sep 13;19(1):136. [PMC free article] [PubMed]
22. Gjika E, Beaulieu JY, Vakalopoulos K, Gauthier M, Bouvet C, Gonzalez A, Morello V, Steiger C, Hirsiger S, Lipsky BA, Uçkay I. Two weeks versus four weeks of antibiotic therapy after surgical drainage for native joint bacterial arthritis: a prospective, randomised, non-inferiority trial. Ann. Rheum. Dis. 2019 Aug;78(8):1114-1121. [PMC free article] [PubMed]
23. Burkhart HM, Deschamps C, Allen MS, Nichols FC, Miller DL, Pairolero PC. Surgical management of sternoclavicular joint infections. J. Thorac. Cardiovasc. Surg. 2003 Apr;125(4):945-9. [PubMed]
24. Sewell MD, Al-Hadithy N, Le Leu A, Lambert SM. Instability of the sternoclavicular joint: current concepts in classification, treatment and outcomes. Bone Joint J. 2013 Jun;95-B(6):721-31. [PubMed]
- ↑ 1.0 1.1 1.2 1.3 1.4 Higginbotham TO, Kuhn JE. Atraumatic disorders of the sternoclavicular joint. J Am Acad Orthop Surg. 2005;13(2):138-45.
- ↑ 2.0 2.1 Logan C, Shahien A, Altintas B, Millett PJ. Rehabilitation Following Sternoclavicular Joint Reconstruction for Persistent Instability. Int J Sports Phys Ther. 2018;13(4):752-62.
- ↑ Spencer EE, Kuhn JE, Huston LJ, Carpenter JE, Hughes RE. Ligamentous restraints to anterior and posterior translation of the sternoclavicular joint. J Shoulder Elbow Surg. 2002;11(1):43-7.
- ↑ Giphart JE, Brunkhorst JP, Horn NH, Shelburne KB, Torry MR, Millett PJ. Effect of plane of arm elevation on glenohumeral kinematics: a normative biplane fluoroscopy study. J Bone Joint Surg Am. 2013;95:238-45.
- ↑ 5.0 5.1 5.2 5.3 5.4 5.5 5.6 Ross JJ, Shamsuddin H. Sternoclavicular septic arthritis: review of 180 cases. Medicine (Baltimore). 2004;83:139-48.
- ↑ 6.0 6.1 Bar-Natan M, Salai M, Sidi Y, Gur H. Sternoclavicular infectious arthritis in previously healthy adults. Semin Arthritis Rheum. 2002;32:189-95.
- ↑ 7.0 7.1 Nusselt T, Klinger HM, Freche S, Schultz W, Baums MH. Surgical management of sternoclavicular septic arthritis. Arch Orthop Trauma Surg. 2011;131:319-23.
- ↑ Brancós MA, Peris P, Miró JM, Monegal A, Gatell JM, Mallolas J, Mensa J, García S, Muñoz-Gomez J. Septic arthritis in heroin addicts. Semin Arthritis Rheum. 1991;21:81-7.
- ↑ 9.0 9.1 9.2 Jain A, Jajodia N, Aggarwal A, Singh J, Gupta S. Tuberculosis of the sternoclavicular joint. J Orthop Surg (Hong Kong). 2015;23:315-8.
- ↑ 10.0 10.1 10.2 von Glinski A, Yilmaz E, Rausch V, Koenigshausen M, Schildhauer TA, Seybold D, Geßmann J. Surgical management of sternoclavicular joint septic arthritis. J Clin Orthop Trauma. 2019;10:406-13.
- ↑ Jang YR, Kim T, Kim MC, Sup Sung H, Kim MN, Kim MJ, Kim SH, Lee SO, Choi SH, Woo JH, Kim YS, Chong YP. Sternoclavicular septic arthritis caused by Staphylococcus aureus: excellent results from medical treatment and limited surgery. Infect Dis (Lond). 2019;51:694-700.
- ↑ 12.0 12.1 Zanelli G, Sansoni S, Migliorini L, Donati E, Cellesi C. Sternoclavicular joint infection in an adult without predisposing risk factors. Infez Med. 2003;11:105-7.
- ↑ 13.0 13.1 Renoult E, Lataste A, Jonon B, Testevuide P, Kessler M. Sternoclavicular joint infection in hemodialysis patients. Nephron.1990;56:212-3.
- ↑ Morell DJ, Thyagarajan DS. Sternoclavicular joint dislocation and its management: A review of the literature. World J Orthop. 2016;7:244-50.
- ↑ Rodchuae M, Ruangpin C, Katchamart W. Clinical manifestations, treatment outcomes, and risk factors for sternoclavicular septic arthritis. Rheumatol Int. 2017;37:819-24.
- ↑ Szentkereszty Z, Pósán J, Petö K, Sápy P, Boros M, Takács I, Sz Kiss S. [Surgical management of sternoclavicular joint infections]. Magy Seb. 2007 Jan;60:514-7.
- ↑ Rockwood CA Jr, Groh GI, Wirth MA, Grassi FA. Resection arthroplasty of the sternoclavicular joint. J Bone Joint Surg Am. 1997;79:387-93
- ↑ 18.0 18.1 Chun JM, Kim JS, Jung HJ, Park JB, Song JS, Park SS, Lee HS, Ahn TS. Resection arthroplasty for septic arthritis of the sternoclavicular joint. J Shoulder Elbow Surg. 2012;21:361-6.
- ↑ 19.0 19.1 Kachala SS, D'Souza DM, Teixeira-Johnson L, Murthy SC, Raja S, Blackstone EH, Raymond DP. Surgical Management of Sternoclavicular Joint Infections. Ann. Thorac. Surg. 2016;101:2155-60.
- ↑ Whitlark JD, Kirollos JA, Jackson SM. Simplified Method for Treating Osteomyelitis of the Sternoclavicular Joint. Ann Thorac Surg. 2016;101:1211-2.
- ↑ Opoku-Agyeman J, Matera D, Simone J. Surgical configurations of the pectoralis major flap for reconstruction of sternoclavicular defects: a systematic review and new classification of described techniques. BMC Surg. 2019;19:136.
- ↑ Gjika E, Beaulieu J, Vakalopoulos K, Gauthier M, Bouvet C, Gonzalez A, Morello V, Steiger C, Hirsiger S, Lipsky BA, Uçkay I. Two weeks versus four weeks of antibiotic therapy after surgical drainage for native joint bacterial arthritis: a prospective, randomised, non-inferiority trial. Annals of the Rheumatic Diseases 2019;78:1114-21.
- ↑ Burkhart HM, Deschamps C, Allen MS, Nichols FC, Miller DL, Pairolero PC. Surgical management of sternoclavicular joint infections. J. Thorac. Cardiovasc. Surg. 2003;125:945-9.
- ↑ Sewell MD, Al-Hadithy N, Le Leu A, Lambert SM. Instability of the sternoclavicular joint: current concepts in classification, treatment and outcomes. Bone Joint J. 2013;95-B:721-31.