Shoulder:Rotator Cuff Pathology/Biology of Rotator Cuff Healing
Written by Alexandre Lädermann
Contents
- 1 Bullet Points
- 2 Key Words
- 3 Introduction
- 4 The Healing Response after Rotator Cuff Tear
- 5 The Rotator Cuff Enthesis and its Augmentation Potential
- 5.1 Growth Factors to Augment the Enthesis
- 5.2 Using Platelet Concentrates to Augment the Enthesis
- 5.3 Cell Based Augmentation
- 5.4 Scaffold based Augmentation
- 6 Augmentation of the Rotator Cuff Muscle
- 7 Conclusion
Bullet Points
- Despite advances in surgical reconstruction of chronic rotator cuff tears leading to improved clinical outcomes, failure rates of 13-94% have been reported. Reasons for this rather high failure rate include compromised healing at the bone-tendon interface, as well as the musculotendinous changes that occur after RC tears, namely retraction and muscle atrophy, as well as fatty infiltration.
- It is important to note that instead of a physiological enthesis, an abundance of scar tissue is formed during healing process.
- Even though cytokines have demonstrated the potential to improve rotator cuff healing in animal models, there is little information about the correct concentration and timing of the more than 1500 cytokines that interact during the healing process.
- There is only minimal evidence that platelet concentrates may lead to improvement in radiographic, but not clinical outcome.
- Using stem cells to biologically augment the reconstruction of the tears might have a great potential since these cells can differentiate into various cell types that are integral for healing.
Key Words
Biological augmentation, Rotator cuff healing, Stem cells, Growth factors, Scaffolds, Shoulder surgery, Platelet rich fibrin, Platelet concentrates, Mesenchymal stem cells, Fatty infiltration, Atrophy, Retraction.
Introduction
Pathologies of the rotator cuff are by far the most common cause of shoulder dysfunction and pain. In the presence of full thickness rotator cuff tears, rotator cuff reconstruction is a commonly performed surgical solution. Even though rotator cuff repair results in improved clinical outcome, several studies report failure of healing in up to 94% of patients.[1]
The reason for these high failure rates may be due to intrinsic degenerative changes of the musculotendinous unit. Subsequent to a tear, the muscle retracts, but this muscular retraction is significantly less then the degree of tendon retraction that occurs at later stages.[2][3]
In the tendon low cellularity, degenerative changes and poor blood supply of the enthesis are significant in the findings. Whilst in the muscle, there is significant migration of inflammatory cells within the first few days of a tear and the muscle fibers undergo apoptosis. In the ensuing weeks to months, this early response leads to muscular retraction, degeneration and atrophy. The progressive loss of muscle volume is due to a loss of sarcomeres in series that is associated with an increase in pennation angle which causes an enlargement of the inter- and intramyofibrillar spaces. If the muscle remains unloaded and retracted, the myogenic precursor cells may be reprogrammed into the adipogenic pathway, with mature adipocytes infiltrating the free inter- and intramyofibrillar spaces. This phenomenon is termed fatty infiltration.[4]
The Healing Response after Rotator Cuff Tear
The healing process is divided into three overlapping stages: inflammation (0-7 days), repair (5–25 days), and remodeling (>21 days). In the initial inflammatory phase, various cytokines released by the injured tissue attract inflammatory cells. These cells release other cytokines such as Interleukin1-ß (IL1-ß) and Tumor Necrosis Factor α (TNFα), that incite the inflammatory cascade. These factors activate Nuclear Factor kappa B (NF-κB), which not only induces apoptosis in the musculotendinous unit but also causes muscle atrophy. Furthermore, NF-κB inhibits the regeneration pathway.[5][6][7][8]
The unloaded musculotendinous unit post-tearing leads to the release and activation of pro-fibrotic factors from the surrounding extracellular matrix (ECM). These factors are members of the Transforming Growth Factor beta (TGFβ) superfamily and are key regulators of gene expression in homeostasis. This early response to rotator cuff tear leads to apoptosis of tenocytes and degradation of muscle fibers. It allows cellular debris to be cleared and subsequent tissue regeneration to occur. Vasoactive factors are released initiating angiogenesis and chemotactic factors are released stimulating cell proliferation. Once the cellular debris has been evacuated, the monocytes get transformed to support new tissue formation.[9]
In the muscle, these anti-inflammatory macrophages express myogenic regulatory factors, which in combination with other endocrine growth factors instigate the development mature myocytes from precursor cells. In the tendon-bone interface, these anti-inflammatory macrophages seem to increase scar tissue formation rather than normal tendon tissue. This scar tissue initially consists of collagen type III. Subsequently, collagen type III is replaced by collagen type I, and therefore the collagen type I to III ratio increases. The complex interplay of molecular and cellular mechanisms at the level of the enthesis, as well as in muscle, leads to further scar tissue formation at the enthesis and irreversible structural alterations in the rotator cuff. Research has therefore focused on altering this scar tissue response using different approaches.[10][11][12]
Biological factors that may influence healing
Several patient specific factors have shown to influence the healing of the rotator cuff. The fact that increasing age may alter rotator cuff healing after rotator cuff repair has been reported by several authors. Oh et al. reported that age was related with poor postoperative integrity in univariate analysis. Thus, multivariate regression showed that age was not an independent determinant for anatomical as well as functional outcome. The only independent predictors found in this study were tear retraction and fatty infiltration. Even though increasing age has shown to negatively impact healing, several studies have reported good outcomes after rotator cuff repair in older patient. Apart of age, several factors that can be influenced by medication or change of lifestyle have been found.[13]
Smoking
Galatz et al. demonstrated that nicotine impairs biomechanical as well as histological properties after rotator cuff tendon repair in a rat model. In a clinical study, a dose- and time-dependent relationship between smoking and the presence of rotator cuff tears was noted. These data suggest that abstinence or at least a decrease in nicotine use might help to improve healing after rotator cuff repair.[14][15]
Diabetes Mellitus
Diabetes may have an impact on rotator cuff healing. Bedi et al. reported that diabetes mellitus decreased biomechanical properties in a rat model. Chen et al. reported a higher rate of postoperative complications, namely infections and to a lesser extent also failures.[16][17]
Use of Nonsteroidal Anti-Inflammatory Drugs (NSAID)
There is some basic research evidence that the application of NSAIDs postoperative may alter rotator cuff healing. The common practice of administering NSAIDs should therefore be reconsidered during the first six postoperative weeks. After this period of time, NSAIDs do not seem to have an influence on healing and there is evidence that they positively influence the remodeling of collagen matrix during that time.[18]
Hypercholesterolemia
There seems to be a relationship between an individual's lipid levels and tendon pathologies. Therefore the question rises, if high serum cholesterol levels should be treated before rotator cuff surgery.[19]
Vitamin D Deficiency
Even though animal studies suggest that low levels of vitamin D may negatively influence early healing at the rotator cuff repair site, in a clinical setting no correlation was found between the severity of rotator cuff tear or the retear rate. Recommendations for clinics are depicted in Table 1 concerning these risk factors.[20][21]
Table 1. Several factors that can be influenced by medication or change of lifestyle have been found. Recommendations for clinics are listed in this table.
The Rotator Cuff Enthesis and its Augmentation Potential
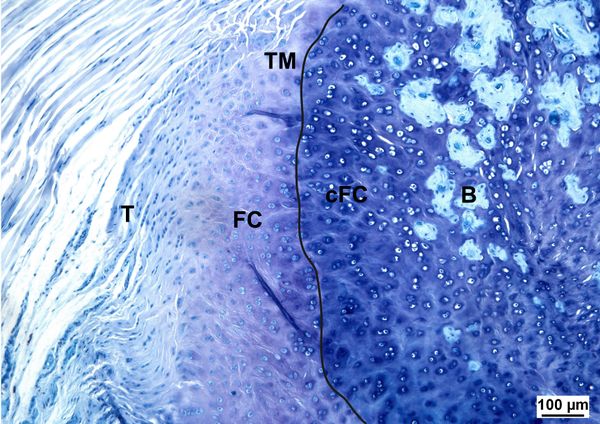
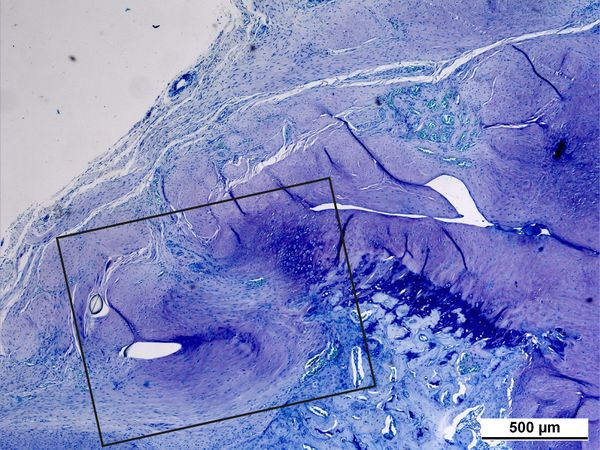
Rotator cuff tears occur predominantly in the enthesis; the transition zone between the tendon and the bone. The tendon-bone junction is divided into four zones, namely tendon, unminerealized fibrocartilage, mineralized fibrocartilage, and bone (Figures 1, 2).[22][23]
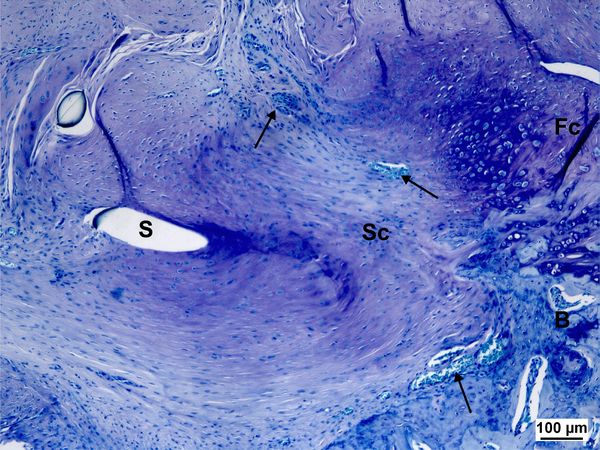
The ensuing scar tissue formation after repair (Figures 3, 4) has a detrimental effect on the biomechanical characteristics of this area compared to the four zones of the physiological enthesis. Hence, there is a strong drive to investigate the role of potential molecular therapies in the form of biological augments that may restore the pathologic enthesis to a near normal physiological state.
Growth Factors to Augment the Enthesis
Several growth factors have been shown to play an integral role in enthesis healing by regulating inflammation and matrix synthesis, as well as cell migration, proliferation, differentiation. The application of a single growth factor has been investigated in vitro and in several animal studies (e.g. BMP-12 or TGFβ). Single growth factor administration did not improve the functional or mechanical properties of the repair. Since the healing process is regulated by more than one growth factor, Rodeo et al implanted TGFb1 to TGFb3, BMP 2 to BMP 7 and FGF applied into a collagen type I sponge in between the tendon and the footprint of a sheep rotator cuff repair healing site.[24]
They reported improved biomechanical properties after 6 and 12 weeks with an improved bone and soft tissue volume compared to the control group, where only a surgical reconstruction was performed. When normalizing the tissue volume of the scar tissue to the normal physiological tendon size, no differences were seen with respect to maximal load to failure. Even though the growth factor cocktail accelerated healing, it did not change quality of repair. To date, there are no clinical studies in humans investigating the role of biological augments with rotator cuff reconstruction.
Using Platelet Concentrates to Augment the Enthesis
Many of the cytokines present during enthesis healing are also found in high concentrations in platelet concentrates (e.g. TGFβ1, PDGF, bFGF, VEGF, EGF, and IGF-1). Platelet concentrates have also been shown to promote neovascularization, which may increase blood supply enabling cells to migrate to the healing site. Since chronic rotator cuff tears show a rather unfavorable healing milieu, neovascularisation may be potentially therapeutic. This hypothesis is partly negated by one study, in which increased neovascularization after biological augmentation of tears using platelet concentrates did not lead to an improved clinical or radiographical outcome.[25]
In contrast to the application of growth factors, autologous platelet concentrates are available without the risk associated with allogenic products and they can be prepared during reconstruction. Platelet concentrates have therefore been explored as a promising potential biologic augment for healing. Preclinical in vitro studies have shown promising results, but only a few large-scale clinical studies have investigated its benefit for the healing rotator cuff with inconclusive results.
The interpretation of the results of these studies is complicated by the fact that there are different platelet concentrates, with varying growth factor release patterns over time. Reasons for these different properties may be found in the lack of standardization between the different platelet concentration systems, with every system containing different concentration of factors and varied activation status of the platelets. Other reasons for inconsistencies between these systems may be due to the different concentrations of other bioactive ingredients such as leukocytes, red blood cells and the fibrinogen. For this reason, Dohan et al. have divided the different platelet concentrates depending on their leukocyte and fibrin content into four categories (Table 1):[26]
- Leucocyte- and platelet-rich plasma (L-PRP) (e.g. GPS III, Magellan and SmartPReP)
- Pure platelet-rich plasma (P-PRP) (e.g. Vivostat PRF, ACP and cell separator PRP)
- Leucocyte and platelet rich fibrin (L-PRF) (e.g. L-PRF)
- Pure platelet-rich fibrin (P-PRF)(e.g. Cascade FIBRINET PRFM System)
Table 2. Platelet-rich concentrates can be divided into four categories depending on their leukocyte and fibrin content. Each group shows different characteristics. The most important ones are depicted in the table.
Leukocyte and platelet-rich plasma (L-PRP)
Full Thickness rotator cuff tears
Several clinical studies have investigated the application of L-PRP to the reconstruction site. Randelli et al. performed a repair in 53 subjects with 26 of these patients receiving L-PRP augmentation. In the control group (n=27), the rotator cuff was reconstructed without a L-PRP treatment. In the first postoperative month, the L-PRP group showed decreased pain scores and after 3 months an increased simple shoulder test (SST). After 6, 12, and 24 months, no radiographical or clinical differences were reported.[27]
Another study, where large and massive tears were augmented using L-PRP also showed no benefit of the L-PRP augmentation except radiographically for smaller iterative tears in the L-PRP group seen in MRI scans. However the clinical outcome measured with the UCLA score, Simple Shoulder Test score, Constant Score and strength was not different in the L-PRP and control group.[28]
One explanation for these results may be the rather high platelet concentrations. The platelet concentrations in L-PRP are up to 4 times higher than in physiological blood. There is evidence that platelet concentrations over 2.5-fold result in a reduction in cell proliferation and might therefore have a negative impact on healing.[29]
Gumina et al. therefore applied platelet-leukocyte membranes, that only allowed 1.7 times greater platelet counts compared to the physiological blood concentrations. They reported improved repair integrity in the L-PRP group compared to the control group, where only a conventional repair was performed (p=.04). However, they did not find any difference with respect to the clinical outcome.[30]
In a recent study, Carr et al. investigated both the clinical and tissue effects of the coapplication of PRP injection with arthroscopic acromioplasty in patients with chronic rotator cuff tendinopathy. Patients were randomized to arthroscopic acromioplasty alone or in combination with an injection of autologous L-PRP into the subacromial bursa. The coapplication of L-PRP did not affect clinical outcomes. Of concern was the finding that L-PRP significantly alters the tissue characteristics in tendons after surgery, with reduced cellularity and vascularity, and increased levels of apoptosis with an increased expression of p53-positive apoptotic cells.[31]
Partial Thickness rotator cuff tears
L-PRP injections within interstitial supraspinatus tears did not improve tendon healing or clinical scores compared to saline injections and are associated with more adverse events.[32]
Pure platelet-rich plasma (P-PRP)
Full Thickness rotator cuff tears
In a published randomized, single blinded study, 48 patients were randomly assigned to receive either a P-PRP augmented (24 patients) or a conventional (24 patients) rotator cuff reconstruction after large or massive tears. The authors used a leucoreduction plasmapheresis system for the production of P-PRP. The retear rate of the P-PRP group (20.0%) was significantly lower than that of the conventional group (55.6%) (P = .023). Clinical outcomes showed no statistical difference between the 2 groups (all P > .05) except for the overall function (P = .043). The change in the 1-year postoperative and immediately postoperative cross-sectional area (CSA) of the supraspinatus was significantly different between the 2 groups: -15.54 ± 94.34 mm² in the PRP group versus -85.62 ± 103.57 mm² in the conventional group (P = .047). A limitation of this study is the rather small sample size.[33]
These promising results were confirmed in another cohort study.[34]
In contrast, no differences were seen in the clinical outcome, nor in the retear rate up to 24 months postoperatively using the Vivostat system.[35]
Again, a limitation of this study is the rather small sample size. Similarly, no significant differences were found when applying Autologous Conditioned Plasma or platelet rich plasma prepared by apharesis during reconstruction in two randomized controlled studies.[36][37]
Another option of augmenting rotator cuff reconstruction is the postoperative injection of platelet concentrates. Autologous conditioned plasma injected in an ultrasound guided manner 7 and 14 days postoperative after reconstruction did not accelerate early tendon healing measured in MRI scans and functional recovery, measured in Quick DASH, Oxford Shoulder Score and VAS compared to a control group.[38]
Partial Thickness rotator cuff tears
Except for a case report, there is no study investigating the impact of P-PRP on the clinical and radiological outcome of partial thickness tears.
Leukocyte and platelet-rich fibrin (L-PRF)
Full Thickness rotator cuff tears
In-vitro studies showed more constant and longer growth factor release patterns in L-PRF compared to L-PRP and better migration properties of mesenchymal stem cells and entdothelial cells.[39]
But to date the clinical studies have not demonstrated any benefits with respect to the use of L-PRF. Zumstein et al. augmented the rotator cuff reconstruction in patients using L-PRF in a randomized controlled trial.[25]
In the 10 patients, where L-PRF was added, an increased vascularization was reported six weeks postoperatively (p= .001) when compared to a control group of patients (n=10), which did not receive L-PRF. Clinical examinations including subjective shoulder value, visual analog scale, Constant, and Simple Shoulder Test scores did not reveal significant differences 6 and 12 weeks postoperatively. A limitation of this study is the rather small sample size. In a subsequent study, 35 patients were randomized to receive arthroscopic reconstruction with L-PRF locally applied to the repair site (n = 17) or without L-PRF (n = 18). They concluded that arthroscopic reconstruction with application of L-PRF yields no beneficial effect in clinical outcome, anatomic healing rate, mean postoperative defect size, and tendon quality at 12 months of follow-up.
Partial Thickness rotator cuff tears
L- PRF is not applicable in partial rotator cuff tears.
Pure platelet-rich fibrin (P-PRF)
Full Thickness rotator cuff tears
Several authors did not show significant differences in clinical outcome and similar or worst radiographical outcome after application of P-RFM.
In a randomized controlled trial, Rodeo et al. performed a rotator cuff reconstruction in 79 patients. The patients were randomized to either receive P-PRF at the tendon-bone interface (n=40) or a standard repair with no P-PRF (n=39). There were no differences in tendon-bone healing rate after 12 months (67% in the P-PRF group, 81% in the control group, p=0.2), the manual muscle strength or the clinical outcome between the two groups. Interestingly, the platelet count had no effect on healing. Regression analysis suggested that P-PRF may have a negative effect on healing (odds ratio: 5.8), as it was a significant predictor for a tendon defect at 12 weeks.[40]
In another cohort study, similar results were found with a significantly higher retear rate in the P-RFM group.[41]
Castricini et al. reported no significant difference when comparing the Constant Score (p=0.44) and the rerupture rate (1 of 40 in the P-PRF group, 4/38 in the control group; p=0.07) of the P-PRF and the control group. Also, no difference was found between both groups when comparing the tendon thickness (p=0.18). However, their results are only applicable for small and medium rotator cuff tears.[42]
Partial Thickness rotator cuff tears
To our knowledge, there is no study investigating the impact of P-PRP on the clinical and radiological outcome of partial thickness tears.
In conclusion, the benefit of platelet concentrates in a clinical setting remains questionable when looking at the current evidence. In a meta-analysis of randomized controlled trials up to 2014, Li et. al. concluded that platelet concentrates have no benefit with respect to retear rate and overall clinical outcomes for the arthroscopic reconstructions.[43]
Cell Based Augmentation
Cells are essential for the healing process of the rotator cuff tears. Tendons are relatively hypocellular. Furthermore, a study by Hernigou et al. has revealed a massive reduction of mesenchymal stem cell levels of up to 70% in bone marrow harvested at the tendon-bone interface of patients with symptomatic tears compared with patients with an intact rotator cuff. The relative lack of cellular machinery in an around the enthesis coupled with poor vascularity results in poor healing potential following acute or overuse injuries[105]. In recent years, biological augmentation using cell-based therapy has been investigated. The application of stem cells has shown promising results.[44]
Stem Cells to Augment the Enthesis
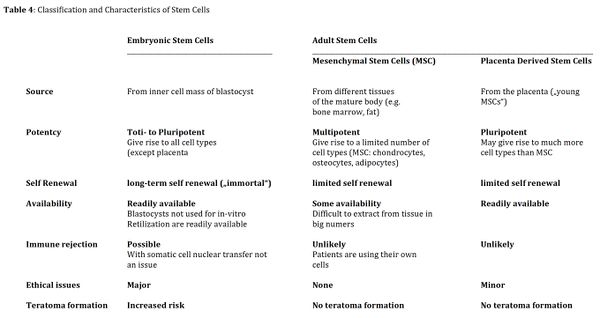
The application of stem cells to the healing site has significant potential as a future therapy. Stem cells can be classified according to their ability to differentiate into other cell types. They can be divided into (1) embryonic stem cells and (2) adult stem cells (Table 3). While totipotent embryonic stem cells from the first couple of cell division cycles can divide into every cell of an organism, pluripotent stem cells lack differentiation into extra-embryonic tissue like the placenta. In contrast, multipotent embryonic stem cells can only divide into a limited range of cells of a single tissue type. Adult stem cells, the second group of stem cells, are multipotent undifferentiated cells found in adult tissue. They include Adipose-derived stem cells (AD-MSC) and Mesenchymal stem cells (MSCs). These cells replace the dying cells in the tissue. Placenta derived stem cells also belong to the adult stem cell group. Unlike MSCs and AD-MSCs, they are pluripotent and have therefore the capacity to differentiate toward all three germ layers.[45]
Table 3. Classification and characteristics of stem cells
Even though embryonic stem cells offer a wider therapeutic potential than adult stem cells, especially when using totipotent cells, their use in research is ethically controversial and an increased risk for teratoma development has been reported. When using adult stem cells ethical issues are not as significant and malignant transformations are much less likely.
Application of mesenchymal stem cells in rotator cuff repair
There is evidence that mesenchymal stem cells may improve rotator cuff repair healing. The possibility to harvest bone marrow aspirate concentrate during reconstruction in a more or less standardized manner and the fact that bone marrow aspirate concentrate apart from other cells and platelets, contains mesenchymal stem cells has made bone marrow aspirate concentrate a promising option for a biological augmentation of the repair.
There is limited literature investigating the clinical effect of the biological augmentation using mesenchymal stem cells or bone marrow aspirate concentrate to improve healing. In a study published in 2014, Hernigou et al. showed that arthroscopic reconstructions that were augmented with bone marrow derived mesenchymal stem cells showed a 13% retear rate 10 years postoperatively compared to 46% in the control group, that only received a rotator cuff single row repair without application of mesenchymal stem cells.[46]
In another study 14 patients with complete rotator cuff tears received a reconstruction with a subsequent injection of autologous bone marrow derived stem cells. The MRI analysis, that was performed 12 months after surgery revealed good tendon integrity in all the subjects. Clinical findings remained unaltered in the following year in all but one patient, who relapsed into loss of strength and pain, considered as a bad result. However, a limitation of this study was the absence of a control group and the rather small study population.[47]
Several studies have investigated the isolation and characterization of mesenchymal stem cells harvested from different shoulder tissues. Mazzocca et al. were able to harvest mesenchymal stem cells from the bone marrow through the anchor tunnel of the humeral head during arthroscopic reconstructions. These cells were then cultivated. Doing so, the authors were able to produce connective tissue progenitor cells, which have the potential of being used in future operations.[48]
In another study, mesenchymal stem cells that were harvested from the humerus were treated with a single physiologic dose of insulin. The authors were able to show that these cells differentiated into cells with characteristics consistent with tendon. The potential for mesenchymal stem cells to differentiate into tendon after a single dose of insulin may assist in developing practical biologic options for augmentation of reconstructions.[49]
Another group characterized mesenchymal stem cells from four different shoulder tissues (synovium of the glenohumeral joint, subacromial bursa, margin of the ruptured supraspinatus tendon, and residual tendon stump on the greater tuberosity) in 19 patients. The subacromial tissue showed more passage 0 cells and these cells kept their proliferative ability for more passages. They also showed a higher osteogenic and adipogenic potential. But the chondrogenic potential in subacromial mesenchymal stem cells was lower in comparison to the mesenchymal stem cells harvested from the enthesis. An interpretation of these in-vitro results is difficult and the conclusion made by the authors was that subacromial bursa mesenchymal stem cells are a good candidate for the source of mesenchymal stem cells in rotator cuff reconstructions. But this is questionable. Not a high adipogenic, but a chondrogenic potential would be desirable for the healing of the enthesis since two out of the four physiological zones in the rotator cuff enthesis consist of fibrocartilage. But this very chondrogenic potential is rather low in the subacromial bursa mesenchymal stem cells.[50]
Application of adipose-derived stem cells in rotator cuff reconstructions
Another source of mesenchymal stem cells is the fatty tissue. Adipose derived stem cells can be harvested via relatively minimally invasive liposuction. When compared to bone marrow derived stem cells, they show a similar morphology, and CD surface marker protein expression, but a higher colony-forming and adipogenic potential.[51]
When injected into a chronic rotator cuff healing model in a rabbit, adipose-derived stem cells increased the maximal load to failure compared to the control group where only saline was injected into the repair site.[52]
However, this difference was not statistically significant. Significantly better tendon architecture, decreased inflammatory cell numbers, and significantly increased tensile strength were found, when human adipose-derived stem cells were injected into a rat rotator cuff tendinopathy model.[53]
In contrast, the application of adipose-derived stem cells in a rat reconstruction model did not improve the biomechanical properties of the tendon-to-bone healing in another study. Due to the contradicting literature, it is unclear if adipose-derived stem cells represent a good alternative to increase the tendon-bone healing in the tear in humans. More animal and human clinical studies will be necessary to prove its benefit in rotator cuff healing.[54]
Application of placental derived stem cells in rotator cuff reconstructions
In the recent years, placental human derived mesenchymal stem cells gained attention in orthopaedic research. Placental human derived mesenchymal stem cells can be isolated and expanded relatively easy and have shown to have multilineage differentiation potential similar to mesenchymal stem cells derived from bone marrow.[55]
In a rat patellar tendinopathy model, the application of placenta derived stem cells has shown better biomechanical, as well as better histomorphological results. This makes these cells attractive for the biological augmentation of the reconstructions. Further in-vitro and future clinical studies will be necessary to explore their safety and potential in rotator cuff healing.[56]
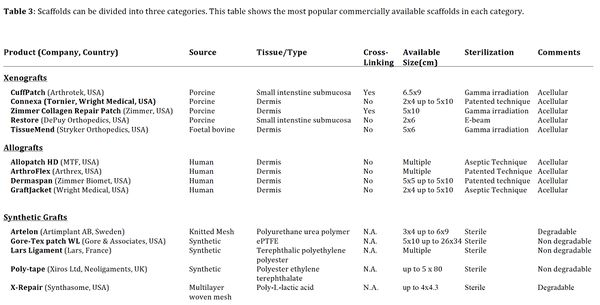
Scaffold based Augmentation
The application of scaffolds is another promising way of augmenting reconstruction sites, especially when dealing with large and complex rotator cuff tears. Even though a scaffold interposition may help bridging rotator cuff tendon defects, it does not address the issues related to rotator cuff retraction, namely the reduced irreversible biomechanical properties of the rotator cuff muscle. The use of interposition scaffolds in rotator cuff repair is therefore questionable. On the other hand, scaffolds with which the tendon is augmented not only may increase the initial strength of the reconstruction and allow gradual tissue ingrowth, but also protect the healing tissue. Three different types of scaffolds exist (Table 4):
1. Xenografts (graft transferred from another species)
2. Allografts (graft from an individual to another of the same species)
3. Synthetic Grafts
Table 4. Scaffolds can be divided into three categories. This table shows the most popular commercially available scaffolds in each category.
While xenografts and allografts are postulated to provide a structural environment that may improve healing as well as remodeling, synthetic grafts lack this feature. But, the mechanical strength of synthetic grafts may help to stabilize the repair until the tissue is strong enough again to transmit the muscle load to the humeral head.
Using Xenografts to Augment the Enthesis
Extracellular matrix patches are scaffold devices designed to create a cellular reaction that leads to an inflammatory response, host cell infiltration, and tendon-like remodeling. Even though the augmentation using porcine small intestine submucosa has shown to improve the histological healing properties (e.g. tendon-like remodeling) in several animal studies, clinical studies, where large to massive chronic rotator cuff tears were augmented using a similar scaffold showed no differences in the clinical outcome when compared to a control group.[57][58]
Another group reported that 4 out of 19 (21%) patients treated with the same xenograft had a severe postoperative inflammatory reaction, which made a reoperation with a debridement and removal of the graft necessary. Since all the intraoperative cultures were negative, this reaction was most likely caused by the xenograft itself. Furthermore, no clinical benefit could be shown after a 2 year follow-up.[59]
Inconsistent results were reported when using Permacol, an acellular porcine dermal collagen matrix. It is cross-linked and thus not susceptible to enzymatic degradation. Badhe et al. reported good results 4.5 years after augmenting massive tears in 10 patients with an intact repair in eight, and a retear in only two patients. On the other hand, Soler et al. were using Permacol as a bridging device. All of the four reconstructions failed within six months postoperatively and had signs of inflammation. No differences were seen when using extracellular matrix scaffolds in open revision surgery.[60][61][62]
More promising results were reported in a study of 9 patients with massive, otherwise unrepairable rotator cuff tears, that have received a porcine dermal collagen patch augmentation. After a minimum follow-up of 2.5 years, all cases showed complete functional recovery as well as covering of the humeral head. Another prospective comparative study showed a reduction of the retear rate and improved clinical outcome scores after ECM augmentation of arthroscopic repairs of large to massive tears.[63][64]
Using Allografts to Augment the Enthesis
When using GraftJacket allograft acellular human dermal matrix for the augmentation of large to massive tears, good results were reported. In a prospective, randomized study, an arthroscopic reconstruction of tears larger than 3 cm was performed with either a GraftJacket augmentation (n= 20) or without augmentation (n= 22). After a follow up of 12 to 24 months (mean 14.5 months), significantly more intact repairs were found in the GraftJacket group (85%) compared to the control group (40%). American Shoulder and Elbow Surgeons and Constant scores in the GraftJacket group were statistically better at last follow-up.[65][66]
Using Synthetic Scaffolds to Augment the Enthesis
Synthetic patch augmentation has shown to protect the repair site and reduce friction in the subacromial space. Ciampi et al. were able to show, in a cohort of 152 patients with posterosuperior tears, a significant decrease in retear rate of 17% in the polypropylene patch augmentation group compared to 51% in the repair-only group at 1-year follow-up, as well as increased abduction strength and elevation at 3-year follow-up.[67]
Poly-l-lactic acid bioabsorbable patches designed specifically to reinforce the surgical repair of tendons supported successful repair of large to massive tears in 83% of patients at 12 months after surgery and 78% of patients at 42 months after surgery, with substantial functional improvement.[68]
Promising results were also reported when using a bi-layered, absorbable reinforced poly (4)-hydroxybutyrate scaffold that can be used to reinforce rotator cuff reconstruction. Augmenting the tear using this scaffold lead to improved repair integrity of 96% in 50 patients at the 6 month follow-up with no additional failure at the 1 year follow-up.[69]
Another interesting approach is the combination of a synthetic patch with an autologous long head of the biceps tendon interposition in irreparable tears. A study in 60 patients showed a retear rate at 12 months follow-up of 15% compared to 40% in the control group. It is unclear though, if the synthetic patch, the long head of the biceps tendon or the combination of the two lead to the improved retear rate.[70]
Augmentation of the Rotator Cuff Muscle
Rotator cuff tears have reportedly been associated with muscle atrophy, retraction and fatty infiltration. These changes have been found to be one of the main causes for a poor outcome after reconstruction. In the recent years, researchers have also focused on preventing and reversing these muscular changes.
Platelet Concentrates to Augment the Rotator Cuff Muscle
There is evidence in the literature that the application of platelet concentrates may have a positive effect on muscle healing, improved pain relief, as well as a quicker return to sport. Others reported no differences with respect to time to return to sport or re-injury rate 1 year after injecting a platelet concentrate into the hamstrings after acute hamstring injuries. In all the above studies, acute muscle injuries were treated with platelet concentrate injections. As the mechanism of muscle-injury after tendon tear is different from an acute muscle injury, it is questionable to what extent these results can be transferred to a chronic rotator cuff tear muscle model. Whether platelet rich concentrates improve muscle quality after rotator cuff tear is unclear and not reported in the literature.[71][72]
Cells to Augment the Rotator Cuff Muscle
In an animal study, Oh et al. tenotomised the subscapularis muscle in rabbits. Six weeks postoperatively, they performed a tendon repair and injected adipose derived stromal cells into the subscapularis muscle. A second group, which received saline instead of adipose-derived stem cells, served as a control group. The adipose derived stromal cells group showed a significantly larger compound muscle action potential area than the control group (p=0.029) and a higher load-to-failure (ADSC-group: 87.02 +/- 29.81 N versus saline group: 59.85+/-37.77 N, p=0.085). Furthermore, subscapularis muscles, in which adipose-derived stem cells were injected showed less fatty infiltration with a fat content of only 29% +/- 15% compared to 43% +/- 9% in the control group. To our knowledge, no clinical studies exist that investigate the benefit of cells for the augmentation of the muscle after reconstruction.[52]
Drugs that may Augment Rotator Cuff Muscle
Poly (ADP-ribose) 4 polymerase-1 (PARP-1) has been shown to be a key regulator of inflammation, apoptosis, muscle atrophy, muscle regeneration as well as adipodyte development. In PARP-1 knock-out (PARP-1 KO) mice, a team performed a combined tenotomy of the supra- and infraspinatus muscle and found significant less muscle retraction (p=0.008) and less fatty infiltration (p=0.028) after 12 weeks and a significant decrease in the expression of inflammatory, apoptotic, adipogenic and muscular atrophy genes at both the 1 week and 6 week time points in the PARP-1 KO group compared to the WT group. The PARP-1 KO group also showed a significant decrease in the expression of inflammatory, apoptotic, adipogenic and muscular atrophy genes at both the 1 week and 6 weeks time points. Therefore, PARP-1 is not only a key regulator of muscular deterioration, but may also be an interesting target to improve muscular properties after rotator cuff tear and repair using a PARP-1 inhibitor.[2]
The application of nandrolone decanoate, an anabolic steroid has shown to not only prevent muscle atrophy but also fatty infiltration in animal models making this drug an interesting treatment option for the prevention of muscle degeneration after tear.[73]
It has been proposed that vitamin D blood levels negatively correlate with fatty infiltration after tear and positively correlate with muscle torque in the same patients.[74]
This suggests that a Vitamin D substitution would have a positive impact on muscular changes after tears and repairs. However, in a clinical study, there was no correlation between rotator cuff healing and vitamin D blood levels.[21]
Conclusion
Due to the high retear rate of the rotator cuff muscle after reconstruction, there is a great interest in its biological augmentation. Even though cytokines have shown the potential to improve the healing in animal models, there is little information about the correct concentration and timing of the more than 1500 cytokines that interact during the healing process. Further studies are necessary to identify the right combination, timing and concentration of the cytokines. Tendinopathies and partial-thickness tears may be interesting targets for the biological augmentation of the rotator cuff. Yet we must be aware that animal studies have shown that, instead of a physiological enthesis, an abundance of scar tissue is formed. Using stem cells to biologically augment the reconstruction might have a great potential since these cells can be differentiated into different cell types that are important for the healing process. However, further studies are necessary to understand how to control these stem cells in a safe and efficient way. Finally, more basic research is required to overcome the problems associated with using grafts as well as developing new drugs for the augmentation of rotator cuff reconstruction.
- ↑ Galatz LM, Ball CM, Teefey SA, Middleton WD, Yamaguchi K. The outcome and repair integrity of completely arthroscopically repaired large and massive rotator cuff tears. J bone Joint Surg Am 2004;86-A(2):219-24.
- ↑ 2.0 2.1 Kuenzler MB, Nuss K, Karol A, Schär MO, Hottiger M, Raniga S, Kenkel D, von Rechenberg B, Zumstein MA. Neer Award 2016: reduced muscle degeneration and decreased fatty infiltration after rotator cuff tear in a poly(ADP-ribose) polymerase 1 (PARP-1) knock-out mouse model. J Shoulder Elbow Surg. 2017;26:733-744.
- ↑ Lädermann A, Zumstein M, Kolo F, Grosclaude M, Koglin L, Schwitzguebel A. In Vivo Clinical and Radiological Effects of Platelet-rich Plasma on Interstitial Supraspinatus Lesion: Case Series. Orthop Traumatol Surg Res. 2016;102:977-982.
- ↑ Lundgreen K, Lian OB, Engebretsen L, Scott A. Tenocyte apoptosis in the torn rotator cuff: a primary or secondary pathological event? Br J Sports Med. 2011;45:1035-9.
- ↑ Angeline ME, Rodeo SA. Biologics in the management of rotator cuff surgery. Clin Sports Med. 2012;31:645-63.
- ↑ Andrianjafiniony T, Dupre-Aucouturier S, Letexier D, Couchoux H, Desplanches D. Oxidative stress, apoptosis, and proteolysis in skeletal muscle repair after unloading. American journal of physiology Cell physiology. 2010;299:C307-15.
- ↑ Millar NL, Hueber AJ, Reilly JH, Xu Y, Fazzi UG, Murrell GA, McInnes IB. Inflammation is present in early human tendinopathy. Am J Sports Med. 2010;38:2085-91.
- ↑ Sishi BJ, Engelbrecht AM. Tumor necrosis factor alpha (TNF-alpha) inactivates the PI3-kinase/PKB pathway and induces atrophy and apoptosis in L6 myotubes. Cytokine. 2011;54:173-84.
- ↑ Arnold L, Henry A, Poron F, Baba-Amer Y, van Rooijen N, Plonquet A, Gherardi RK, Chazaud B. Inflammatory monocytes recruited after skeletal muscle injury switch into antiinflammatory macrophages to support myogenesis. The Journal of experimental medicine. 2007;204:1057-69.
- ↑ Frey E, Regenfelder F, Sussmann P, Zumstein M, Gerber C, Born W, Fuchs B. Adipogenic and myogenic gene expression in rotator cuff muscle of the sheep after tendon tear. Journal of orthopaedic research : official publication of the Orthopaedic Research Society. 2009;27:504-9.
- ↑ Zanou N, Gailly P. Skeletal muscle hypertrophy and regeneration: interplay between the myogenic regulatory factors (MRFs) and insulin-like growth factors (IGFs) pathways. Cellular and molecular life sciences : CMLS. 2013;70:4117-30.
- ↑ Hays PL, Kawamura S, Deng XH, Dagher E, Mithoefer K, Ying L, et al. The role of macrophages in early healing of a tendon graft in a bone tunnel. J Bone Joint Surg Am. 2008;90:565-79.
- ↑ Oh JH, Kim SH, Kang JY, Oh CH, Gong HS. Effect of age on functional and structural outcome after rotator cuff repair. Am J Sports Med. 2010;38:672-8.
- ↑ Galatz LM, Silva MJ, Rothermich SY, Zaegel MA, Havlioglu N, Thomopoulos S. Nicotine delays tendon-to-bone healing in a rat shoulder model. J Bone Joint Surg Am. 2006;88:2027-34.
- ↑ Baumgarten KM, Gerlach D, Galatz LM, Teefey SA, Middleton WD, Ditsios K, Yamaguchi K. Cigarette smoking increases the risk for rotator cuff tears. Clin Orthop Relat R. 2010;468:1534-41.
- ↑ Bedi A, Fox AJ, Harris PE, Deng XH, Ying L, Warren RF, Rodeo SA. Diabetes mellitus impairs tendon-bone healing after rotator cuff repair. J Shoulder Elbow Surg. 2010;19:978-88.
- ↑ Chen AL, Shapiro JA, Ahn AK, Zuckerman JD, Cuomo F. Rotator cuff repair in patients with type I diabetes mellitus. J Shoulder Elbow Surg. 2003;12:416-21.
- ↑ Cohen DB, Kawamura S, Ehteshami JR, Rodeo SA. Indomethacin and celecoxib impair rotator cuff tendon-to-bone healing. Am J Sports Med. 2006;34:362-9.
- ↑ Tilley BJ, Cook JL, Docking SI, Gaida JE. Is higher serum cholesterol associated with altered tendon structure or tendon pain? A systematic review. Br J Sports Med. 2015;49:1504-9.
- ↑ Angeline ME, Ma R, Pascual-Garrido C, Voigt C, Deng XH, Warren RF, Rodeo SA. Effect of Diet-Induced Vitamin D Deficiency on Rotator Cuff Healing in a Rat Model. Am J Sports Med. 2014;42:27-34.
- ↑ 21.0 21.1 Ryu KJ, Kim BH, Lee Y, Dan J, Kim JH. Low Serum Vitamin D Is Not Correlated With the Severity of a Rotator Cuff Tear or Retear After Arthroscopic Repair. Am J Sports Med. 2015;43:1743-50.
- ↑ Thomopoulos S, Hattersley G, Rosen V, Mertens M, Galatz L, Williams GR, Soslowsky LJ. The localized expression of extracellular matrix components in healing tendon insertion sites: an in situ hybridization study. Journal of Orthopaedic Research. 2002;20:454–63.
- ↑ Woo SL, Hildebrand K, Watanabe N, Fenwick JA, Papageorgiou CD, Wang JH. Tissue engineering of ligament and tendon healing. Clin Orthop Relat Res. 1999;(367 Suppl):S312-23.
- ↑ Rodeo SA, Potter HG, Kawamura S, Turner AS, Kim HJ, Atkinson BL. Biologic augmentation of rotator cuff tendon-healing with use of a mixture of osteoinductive growth factors. J Bone Joint Surg Am. 2007;89:2485-97.
- ↑ 25.0 25.1 Zumstein MA, Rumian A, Lesbats V, Schaer M, Boileau P. Increased vascularization during early healing after biologic augmentation in repair of chronic rotator cuff tears using autologous leukocyte- and platelet-rich fibrin (L-PRF): a prospective randomized controlled pilot trial. J Shoulder Elbow Surg. 2014;23:3-12.
- ↑ Dohan Ehrenfest DM, Rasmusson L, Albrektsson T. Classification of platelet concentrates: from pure platelet-rich plasma (P-PRP) to leucocyte- and platelet-rich fibrin (L-PRF). Trends Biotechnol. 2009;27:158-67.
- ↑ Randelli P, Arrigoni P, Ragone V, Aliprandi A, Cabitza P. Platelet rich plasma in arthroscopic rotator cuff repair: a prospective RCT study, 2-year follow-up. J Shoulder Elbow Surg. 2011;20:518-28.
- ↑ Charousset C, Zaoui A, Bellaiche L, Piterman M. Does autologous leukocyte-platelet-rich plasma improve tendon healing in arthroscopic repair of large or massive rotator cuff tears? Arthroscopy. 2014;30:428-35.
- ↑ Graziani F, Ivanovski S, Cei S, Ducci F, Tonetti M, Gabriele M. The in vitro effect of different PRP concentrations on osteoblasts and fibroblasts. Clinical oral implants research. 2006;17:212-9.
- ↑ Gumina S, Campagna V, Ferrazza G, Giannicola G, Fratalocchi F, Milani A, Postacchini F. Use of platelet-leukocyte membrane in arthroscopic repair of large rotator cuff tears: a prospective randomized study. J Bone Joint Surg Am 2012;94:1345-52.
- ↑ Carr AJ, Murphy R, Dakin SG, Rombach I, Wheway K, Watkins B, Franklin SL. Platelet-Rich Plasma Injection With Arthroscopic Acromioplasty for Chronic Rotator Cuff Tendinopathy: A Randomized Controlled Trial. Am J Sports Med. 2015;43:2891-7.
- ↑ Schwitzguebel AJ, Kolo FC, Tirefort J, Kourhani A, Nowak A, Gremeaux V, Saffarini M, Lädermann A.Efficacy of Platelet-Rich Plasma for the Treatment of Interstitial Supraspinatus Tears: A Double-Blinded, Randomized Controlled Trial. Am J Sports Med. 2019;47:1885-1892.
- ↑ Jo CH, Shin JS, Lee YG, Shin WH, Kim H, Lee SY, Yoon KS, Shin S. Platelet-rich plasma for arthroscopic repair of large to massive rotator cuff tears: a randomized, single-blind, parallel-group trial. Am J Sports Med. 2013;41:2240-8.
- ↑ Jo CH, Shin JS, Shin WH, Lee SY, Yoon KS, Shin S. Platelet-Rich Plasma for Arthroscopic Repair of Medium to Large Rotator Cuff Tears: A Randomized Controlled Trial. Am J Sports Med. 2015;43:2102-10.
- ↑ Antuna S, Barco R, Martinez Diez JM, Sanchez Marquez JM. Platelet-rich fibrin in arthroscopic repair of massive rotator cuff tears: a prospective randomized pilot clinical trial. Acta orthopaedica Belgica. 2013;79:25-30.
- ↑ Malavolta EA, Gracitelli ME, Ferreira Neto AA, Assuncao JH, Bordalo-Rodrigues M, de Camargo OP. Platelet-rich plasma in rotator cuff repair: a prospective randomized study. Am J Sports Med. 2014;42:2446-54.
- ↑ Werthel JD, Pelissier A, Massin P, Boyer P, Valenti P. Arthroscopic double row cuff repair with suture-bridging and autologous conditioned plasma injection: Functional and structural results. Int J Shoulder Surg. 2014;8:101-6.
- ↑ Wang A, McCann P, Colliver J, Koh E, Ackland T, Joss B, Zheng M, Breidahl B. Do postoperative platelet-rich plasma injections accelerate early tendon healing and functional recovery after arthroscopic supraspinatus repair? A randomized controlled trial. Am J Sports Med. 2015;43:1430-7.
- ↑ Schar MO, Diaz-Romero J, Kohl S, Zumstein MA, Nesic D. Platelet-rich concentrates differentially release growth factors and induce cell migration in vitro. Clin Orthop Relat R. 2015;473:1635-43.
- ↑ Rodeo SA, Delos D, Williams RJ, Adler RS, Pearle A, Warren RF. The effect of platelet-rich fibrin matrix on rotator cuff tendon healing: a prospective, randomized clinical study. Am J Sports Med. 2012;40:1234-41.
- ↑ Bergeson AG, Tashjian RZ, Greis PE, Crim J, Stoddard GJ, Burks RT. Effects of platelet-rich fibrin matrix on repair integrity of at-risk rotator cuff tears. Am J Sports Med. 2012;40:286-93.
- ↑ Castricini R, Longo UG, De Benedetto M, Panfoli N, Pirani P, Zini R, Maffulli N, Denaro V. Platelet-rich plasma augmentation for arthroscopic rotator cuff repair: a randomized controlled trial. Am J Sports Med. 2011;39:258-65.
- ↑ Li X, Xu CP, Hou YL, Song JQ, Cui Z, Yu B. Are platelet concentrates an ideal biomaterial for arthroscopic rotator cuff repair? A meta-analysis of randomized controlled trials. Arthroscopy. 2014;30:1483-90.
- ↑ Hernigou P, Merouse G, Duffiet P, Chevalier N, Rouard H. Reduced levels of mesenchymal stem cells at the tendon-bone interface tuberosity in patients with symptomatic rotator cuff tear. Int Orthop. 2015;39:1219-25.
- ↑ Miao Z, Jin J, Chen L, Zhu J, Huang W, Zhao J, Qian H, Zhang X. Isolation of mesenchymal stem cells from human placenta: comparison with human bone marrow mesenchymal stem cells. Cell biology international. 2006;30:681-7.
- ↑ Hernigou P, Flouzat Lachaniette CH, Delambre J, Zilber S, Duffiet P, Chevallier N, Rouard H. Biologic augmentation of rotator cuff repair with mesenchymal stem cells during arthroscopy improves healing and prevents further tears: a case-controlled study. Int Orthop. 2014;38:1811-8.
- ↑ Ellera Gomes JL, da Silva RC, Silla LM, Abreu MR, Pellanda R. Conventional rotator cuff repair complemented by the aid of mononuclear. Knee Surg Sports Traumatol Arthrosc. 2012;20:373-7.
- ↑ Mazzocca AD, McCarthy MB, Chowaniec DM, Cote MP, Arciero RA, Drissi H. Rapid isolation of human stem cells (connective tissue progenitor cells) from the. Am J Sports Med. 2010;38:1438-47.
- ↑ Mazzocca AD, McCarthy MB, Chowaniec D, Cote MP, Judson CH, Apostolakos J, Solovyova O, Beitzel K, Arciero RA. Bone marrow-derived mesenchymal stem cells obtained during arthroscopic rotator cuff repair surgery show potential for tendon cell differentiation after treatment with insulin. Arthroscopy. 2011;27:1459-71.
- ↑ Utsunomiya H, Uchida S, Sekiya I, Sakai A, Moridera K, Nakamura T. Isolation and characterization of human mesenchymal stem cells derived from shoulder tissues involved in rotator cuff tears. Am J Sports Med. 2013;41:657-68.
- ↑ Vishnubalaji R, Al-Nbaheen M, Kadalmani B, Aldahmash A, Ramesh T. Comparative investigation of the differentiation capability of bone-marrow- and adipose-derived mesenchymal stem cells by qualitative and quantitative analysis. Cell and tissue research. 2012;347:419-27.
- ↑ 52.0 52.1 Oh JH, Song BW, Lee YS. Measurement of volumetric bone mineral density in proximal humerus using quantitative computed tomography in patients with unilateral rotator cuff tear. J Shoulder Elbow Surg. 2014;23:993-1002.
- ↑ Chen HS, Su YT, Chan TM, Su YJ, Syu WS, Harn HJ, Lin SZ, Chiu SC. Human adipose-derived stem cells accelerate the restoration of tensile strength of tendon and alleviate the progression of rotator cuff injury in a rat model. Cell Transplant. 2015;24:509-20.
- ↑ Valencia Mora M, Antuna Antuna S, Garcia Arranz M, Carrascal MT, Barco R. Application of adipose tissue-derived stem cells in a rat rotator cuff repair model. Injury. 2014;45 Suppl 4:S22-7.
- ↑ Fukuchi Y, Nakajima H, Sugiyama D, Hirose I, Kitamura T, Tsuji K. Human placenta-derived cells have mesenchymal stem/progenitor cell potential. Stem Cells. 2004;22(5):649-58.
- ↑ Ma R, Schaer M, Sisto ML, Chen KY, Wang H, E.Z. G, et al. Use of Placental-derived Adherent Stromal Cells Improves Healing in a Preclinical Model of Tendon Injury. American Academy of Orthopaedic Surgeons (AAOS) 2014 Annual Meeting; New Orleans, Louisiana 2014.
- ↑ Nicholson GP, Breur GJ, Van Sickle D, Yao JQ, Kim J, Blanchard CR. Evaluation of a cross-linked acellular porcine dermal patch for rotator cuff repair augmentation in an ovine model. J Shoulder Elbow Surg. 2007;16(5 Suppl):S184-90.
- ↑ Iannotti JP, Codsi MJ, Kwon YW, Derwin K, Ciccone J, Brems JJ. Porcine small intestine submucosa augmentation of surgical repair of chronic two-tendon rotator cuff tears. A randomized, controlled trial. J Bone Joint Surg Am. 2006;88:1238-44.
- ↑ Walton JR, Bowman NK, Khatib Y, Linklater J, Murrell GA. Restore orthobiologic implant: not recommended for augmentation of rotator cuff repairs. J Bone Joint Surg Am. 2007;89:786-91.
- ↑ Badhe SP, Lawrence TM, Smith FD, Lunn PG. An assessment of porcine dermal xenograft as an augmentation graft in the treatment of extensive rotator cuff tears. J Shoulder Elbow Surg. 2008;17(1 Suppl):35S-9S.
- ↑ Sears BW, Choo A, Yu A, Greis A, Lazarus M. Clinical outcomes in patients undergoing revision rotator cuff repair with extracellular matrix augmentation. Orthopedics. 2015;38:e292-6.
- ↑ Soler JA, Gidwani S, Curtis MJ. Early complications from the use of porcine dermal collagen implants (Permacol) as bridging constructs in the repair of massive rotator cuff tears. A report of 4 cases. Acta Orthop Belg. 2007;73:432-6.
- ↑ Gilot GJ, Alvarez-Pinzon AM, Barcksdale L, Westerdahl D, Krill M, Peck E. Outcome of Large to Massive Rotator Cuff Tears Repaired With and Without Extracellular Matrix Augmentation: A Prospective Comparative Study. Arthroscopy. 2015;31:1459-65.
- ↑ Giannotti S, Ghilardi M, Dell'osso G, Magistrelli L, Bugelli G, Di Rollo F, Ricci G, Calabrese R, Siciliano G, Guido G. Study of the porcine dermal collagen repair patch in morpho-functional recovery of the rotator cuff after minimum follow-up of 2.5 years. Surgical technology international. 2014;24:348-52.
- ↑ Barber FA, Burns JP, Deutsch A, Labbe MR, Litchfield RB. A prospective, randomized evaluation of acellular human dermal matrix augmentation for arthroscopic rotator cuff repair. Arthroscopy. 2012;28:8-15.
- ↑ Wong I, Burns J, Snyder S. Arthroscopic GraftJacket repair of rotator cuff tears. J Shoulder Elbow Surg. 2010;19(2 Suppl):104-9.
- ↑ Ciampi P, Scotti C, Nonis A, Vitali M, Di Serio C, Peretti GM, Fraschini G. The benefit of synthetic versus biological patch augmentation in the repair of posterosuperior massive rotator cuff tears: a 3-year follow-up study. Am J Sports Med. 2014;42:1169-75.
- ↑ Proctor CS. Long-term successful arthroscopic repair of large and massive rotator cuff tears with a functional and degradable reinforcement device. J Shoulder Elbow Surg. 2014;23:1508-13.
- ↑ Barbash S, Denny C, Collin P, Reish T, Hart JM, Brockmeier SF. Clinical Outcomes and Structural Healing After Arthroscopic Rotator Cuff Repair Reinforced With A Novel Absorbable Biologic Scaffold: A Prospective, Multicenter Trial. Orthopaedic Journal of Sports Medicine. 2015;3(2 suppl).
- ↑ Vitali M, Cusumano A, Pedretti A, Naim Rodriguez N, Fraschini G. Employment of synthetic patch with augmentation of the long head of the biceps tendon in irreparable lesions of the rotator cuff: our technique applied to 60 patients. Tech Hand Up Extrem Surg. 2015;19:32-9.
- ↑ Wright-Carpenter T, Klein P, Schaferhoff P, Appell HJ, Mir LM, Wehling P. Treatment of muscle injuries by local administration of autologous conditioned serum: a pilot study on sportsmen with muscle strains. Int J Sports Med. 2004;25:588-93.
- ↑ Bubnov R, Yevseenko V, Semeniv I. Ultrasound guided injections of platelets rich plasma for muscle injury in professional athletes. Comparative study. Medical ultrasonography. 2013;15:101-5.
- ↑ Gerber C, Meyer DC, Fluck M, Benn MC, von Rechenberg B, Wieser K. Anabolic Steroids Reduce Muscle Degeneration Associated With Rotator Cuff Tendon Release in Sheep. Am J Sports Med. 2015;43:2393-400.
- ↑ Oh JH, Kim SH, Kim JH, Shin YH, Yoon JP, Oh CH. The level of vitamin D in the serum correlates with fatty degeneration of the muscles of the rotator cuff. J Bone Joint Surg Br. 2009;91:1587-93.