Shoulder:Radiographic Evaluation of Shoulder Problems
Contents
Bullet Points
Key words
Introduction
The shoulder is a complex joint involving a variety of pathologies, whether traumatic or related to a natural aging process. With a population becoming increasingly aging and active, surgery of this joint has grown significantly. Considering this, knowledge of anatomy and its radiological representation is essential in the management of patients with a shoulder problem. Thus, magnetic resonance imaging and computed tomography scans play an important role in the lesion evaluation.[1][2] Small et al. showed that up to 40% of patients who had magnetic resonance imaging for shoulder pain prescribed by a non-specialist did not have a prior conventional radiograph.[3] These magnetic resonance imaging scans seem to have no significant influence on the treatment meaning that a non-specialized physician could overprescribe a magnetic resonance imaging as a screening test. However, in clinical practice, the standard procedure in the assessment of a shoulder pathology is to start with a conventional radiography which, together with the symptomatology, will be the decision-making procedure for further investigations or the treatment. Neer in 1970 described the fractures of the proximal humerus with the surgical indications based on the conventional radiography.[4] In the nineties, we see an emergence of hypotheses concerning the involvement of the scapular anatomy in the tendinopathies of the cuff. Bigliani describes the implication of acromial morphology as a risk factor in rotator cuff injury[5][6] while Hamada describes the arthropathy related to the cuff and its radiological implications.[7] More recently, other authors have described these anatomical implications in rotator cuff disorders and radiological diagnosis.[8][9] Furthermore, understanding the anatomy of the shoulder also involves making surgical decisions according to standard radiography and implant designs.[10][11]
Traumatology
Greater Tuberosity Displacement
The isolated fracture of the greater tuberosity has been described for many years with the most important decision criterion being the displacement measured on the x-ray.[4][12][13][14] In the past, many authors have placed the surgical indication from 1 cm and a conservative treatment below 5 mm.[4][12][13] Although Neer in 1970 described good functional results in conservative treatments for fractures of less than 1 cm with early rehabilitation, there is still a shadow area for fractures with displacement ranging from 5 mm to 1 cm. Understanding the effects of a displacement for this 5-10 mm range becomes clearer. Park et al. describe better functional results when greater tuberosity fixation is performed from a 5 mm displacement or even 3 mm in patients with jobs involving the use of the upper limbs above the head.[15] These results are confirmed by the study of Platzer et al., which shows significantly better functional results with fractures whose displacement is less than 5 mm and which are conservatively treated.[16] He adds, however, that there is a non-significant difference from 3 mm, which could motivate surgery in a particular case. These results can be explained by a defect in abduction due to alteration of the rotator cuff.[12][17] Bono et al. show in an in vitro study that the increase in force required for abduction, when the displacement is more than 5 mm, is statistically significant.[18] This study also reveals a functional deficit when the displacement is posterior. The classification according to Neer does not allow us to judge this displacement. In this context, Mutch et al. describe a classification based on the morphology of fractures that may influence the management of greater tuberosity fractures by classifying them according to whether there is an avulsion, depression or split.[19] They also describe a method of measurement based on radiology to guide the therapeutic decision (Figure).[20] It is therefore essential to measure and analyze the displacement of isolated fractures of the great tuberosity and to include the patient's characteristics in the decision-making strategy in order to avoid functional disorders related to the healing process.
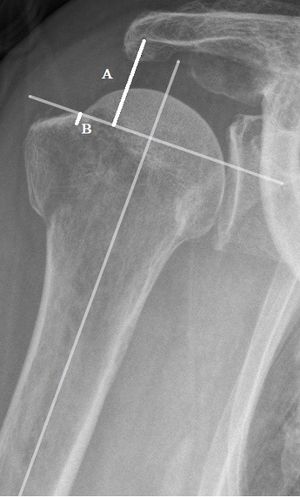
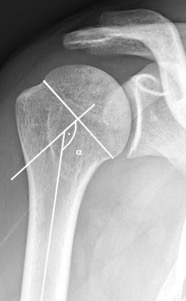
Neck-Shaft angle
The anatomy of the proximal humerus is an essential point in shoulder surgery. In addition to fragments displacement during fractures, the neck-shaft angle is an essential point. Indeed, displacement in valgus or varus during fracture of the proximal humerus can negatively influence the long-term function of the shoulder particularly in a varus positioning being more unstable and of worse prognosis.[21][22][23][24] Restoring the native neck-shaft angulation thus plays an important role in the therapeutic strategy and represents a key factor in the development of surgical techniques for the planning of osteosynthesis or of corrective osteotomy.[10][25] Moreover, the understanding of this angle has made it possible to better adapt prosthetic surgery to the shoulder, particularly in the improvement of implants.[11][26] Thus the normal average value described is 135 degrees (Figure).[27] In the majority of cases, this angle is evaluated on 2D imagery. However, the position of the patient and his shoulder during these examinations may vary from one center to another or even within the same center. Thus, Malatova et al. analyzed angle variations on standard anteroposterior radiography according to the rotation of the shoulder. They show that the angle varies little if the shoulder is in neutral, external or internal rotation with a good correlation between the different observers.[28] On the opposite, other more recent studies contradict this affirmation. Hengg et al. demonstrate that the external and internal rotation of the arm can result in an incorrectly diagnosed valgus.[29] For his part, Adikrishna et al. analyzed the relationships between rotation and NSA with significant differences as early as 10 degrees from RI and 18 degrees from external rotation.[30] It is difficult to judge the rotation by looking at an x-ray, without knowing the positioning of the patient. Tan et al. in a cadaveric study analyze an anatomical reference that can be used to judge the rotation on an x-ray.[31] It shows that the proportional distance of the crest of the small tuberosity from the diameter of the humeral head is about one-third. This proportion decreases with the external rotation and increases with the internal rotation. This would make it possible to judge the rotation on an X-ray or even on intra-operative fluoroscopy. It would therefore appear that standardizing the taking of neutral rotating shots is essential for good practice.
Rotator Cuff Evaluation
X-rays
The analysis should always begin with plain radiographic views to determine the morphology and status of the glenohumeral joint to exclude glenohumeral arthritis.
Anteroposterior
True anteroposterior X-ray with the arm in neutral rotation, and the patient relaxed is obtained to evaluate the shape of the acromion and greater tuberosity, the critical shoulder angle, and the acromiohumeral distance. A decreased acromiohumeral distance < 7 mm in a standard antero-posterior radiograph indicates superior migration of the humeral head which increases the probability of finding an irreparable cuff tear. Such distance is correlated to 1) tears of the infraspinatus that mainly acts in lowering the humeral head, and 2) varying degrees of fatty infiltration.[8][32]
Nevertheless, such criteria should be interpreted with parsimony. First, it is difficult in clinical practice to obtain standardized X-rays making measurement aleatory. Second, this distance has not been associated with an inability to obtain an intra-operative complete repair of the supraspinatus (18.2% irreparable, OR = 0.55, P = 0.610).[33]
At the end of the spectrum, acetabularization of the acromion and femoralization of the humeral head are pre-operative adapting factors reflecting significant chronic static superior instability and are a contraindication for repair.
Lateral Y-view (Lamy)
Lateral Y-view (Lamy) is used to analyze the presence of a spur, the shape of the acromion on this view is less accurate to detect full-thickness rotator cuff tear.[34]
Axillary lateral
An axillary lateral view can exclude static anterior subluxation or os acromialis.
If pathology of the acromioclavicular joint is suspected, a Zanca view is additionally acquired.[35]
Acromial Morphology
The anatomical characteristics of the scapula also have an important implication in the pathologies of the shoulder. The shape of the acromion and its involvement in the lesions of the rotator cuff has been studied by numerous authors.[5][6][36][37] Thus, a link has been demonstrated between the radiological morphology and the risk of degenerative lesion of the rotator cuff, especially the supraspinatus with Bigliani describing 3 types of acromion, namely flat, curved or hooked.[5] It shows an increase in prevalence with progression of type of acromion. However, there is a low inter-observer reliability, especially for types 2 and 3. Other studies confirm this analysis with the absence of objective criteria making the delimitation between type 2 and 3 more complicated.[38] It was in 2001 that Park et al. did a study to give standardized and objective criteria to distinguish the different types based on Neer’s radiological incidence images (Figure).[39] Thus they demonstrate a better inter-class correlation (0.94) with their measurement systems which is more objective and reliable, especially when it is necessary to distinguish type 2 and 3.
Acromial Slope
Another radiological sign based on the morphology of the acromion is described in 1986 by Aoki et al.[40] It describes the influence of the acromial slope on the lesions of the rotator cuff. Thus, a weak acromial slope measured on the Neer’s incidence is an important factor in the subacromial impingement (Figure). Other studies also confirm this trend.[41][42][33]
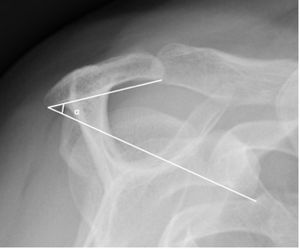
Lateral Acromion Angle
In 1995, Banas et al. introduce the notion of the lateral acromion angle (Figure).[43] In this study, measurements are performed on magnetic resonance imaging coronal sections. It is demonstrated that a low lateral acromion angle is significantly associated with a lesion of the cuff at magnetic resonance imaging with in particular a limit below 70 degrees which would be an indicator for an anterolateral decompression of the acromion. These results are confirmed by a more recent study based this time on standard radiographs of the face.[33] It is also shown that below the threshold of 70 degrees, the lesion of the cuff is constant. Thus this measure could help to guide the therapeutic attitude without complementary exams and help the surgeon in the decision process.
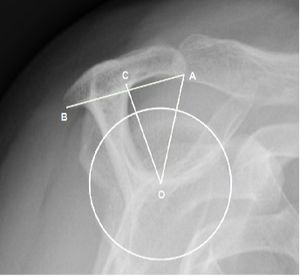
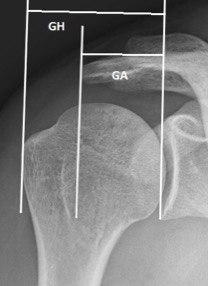
Acromial Index
Recently, Nyffeler et al. were interested in the implication of the extension of the acromion in the tears of the rotator cuff.[8] Their hypothesis was that a broad acromion implies higher deltoid ascending forces which favors impingement and degenerative changes. They therefore describe the radiological measurement of the acromial index which represents the ratio between the glenoid-acromion distance and that between the glenoid-greater tuberosity (Figure). There is therefore an association between a high acromial index and a degenerative lesion of the rotator cuff, thus allowing it to be predicted radiologically. Conversely, they describe an increase in compressive forces on the glenoid when the acromion is short and thus the acromial index low. This increase in compressive force would therefore favor the appearance of glenohumeral arthritis. These findings are confirmed by other studies that also demonstrate an association between elevated acromial index and a tear in the rotator cuff,[44][45] including one also demonstrating higher acromial index in patients with recurrences of postoperative rotator cuff tears.[46] The involvement of the acromial index in the pathologies of the rotator cuff is not however unanimous.[47][48][49] Hamid et al. do not demonstrate any significant association between elevated acromial index and rotator cuff disease,[49] while Kircher et al. do not show an association between a low acromial index and glenohumeral arthritis, which refutes the theoretical concept of a low acromial index resulting in increased contact pressure.[47] Furthermore, Melean et al. do not find correlation between acromial index and the rate of recurrence of rupture of the rotator cuff after surgery. The importance of the acromial index in assessing a shoulder radiograph remains controversial.
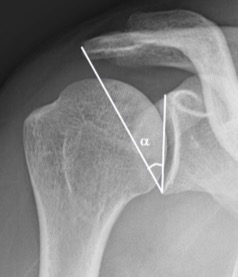
Critical Shoulder Angle
In addition to the morphology or size of the acromion, the variation of the inclination of the glenoid is a radiographic marker that may indicate rotator cuff lesions. Thus, several studies show a relationship between a high glenoid tilt and the upper migration of the humeral head.[50][51] This migration favors subacromial compression of the supraspinatus and therefore tendon tears. In 2013, Moor et al. describe a new radiological marker, the critical shoulder angle.[9] The interest is to include in one measure the concept of the acromial index and the glenoid inclination. It is represented by the angle between a line connecting the upper and lower pole of the glenoid and another connecting the lower pole of the glenoid with the lateral edge of the acromion (Figure). It demonstrates an association between a tear of the cuff and a critical shoulder angle greater than 35 degrees and inversely an association between an osteoarthritis and a critical shoulder angle of less than 30 degrees. Gerber et al. confirm this relationship in a biomechanical study.[52] They show that the increase in critical shoulder angle is associated with an increase in shear forces, especially in degrees of mobility associated with a large number of activities of daily life. The result is a greater antero-superior instability involving more necessary activity on the part of the supraspinatus to stabilize the shoulder and leading more easily to tears. These theories and the association between critical shoulder angle, osteoarthritis and tear of the cuff are also demonstrated significantly in more recent studies.[53][54][55][56][57][58] Engelhardt et al. even independently analyzed the three different parameters (acromial index, IG, critical shoulder angle) demonstrating that critical shoulder angle is the best parameter for estimating the risk of rotator cuff injury.[45] This can be explained by the fact that it combines the influence of a GI and a high acromial index on the upper migration of the humeral head. However, he does not find this precision when it comes to correlating the critical shoulder angle with osteoarthritis. More recently, Chalmers et al. have achieved less satisfactory results.[59] They obtained differences of up to 2 degrees, but too low to have a significant association on the presence of a tendinopathy of the cuff. Since the critical shoulder angle is a radiological measure, it is important to have a good correlation of the measurement. Bouaicha et al. demonstrated in their study that the correlation between anteroposterior standard radiography and scanner is very satisfactory and that the differences that can occur between these two modalities are quite negligible.[60] Spiegl et al. and Cherchi et al. demonstrate a good intra- and inter-observer correlation in the radiological analysis of the critical shoulder angle.[57][58] However, Suter et al. describe the measurement of the critical shoulder angle as a function of the spatial relationship of the scapula as a function of the radiological monitor.[61] They thus note that the critical shoulder angle is sensitive to the ante or retro-version of the scapula relative to the radiological monitor, with a change from 5 to 8 degrees which can change the critical shoulder angle measurement by 2 degrees and thus influence the clinical interpretation.
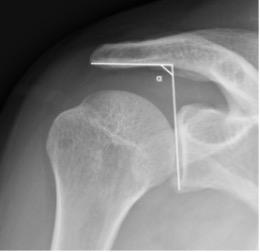
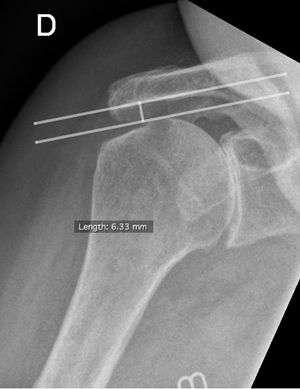
Acromiohumeral Distance
Tendinopathies of the rotator cuff are frequent pathologies which may have as their origin intrinsic and/or extrinsic factors. A magnetic resonance imaging or computed tomography allows these lesions to be visualized with good precision. However, a standard X-ray marker was described long ago. This is the subacromial space (Figure). In the 1960s, Golding was one of the first to describe a link between the decrease in subacromial space and rotator cuff disease.[62] Later in 1970, Weiner et al. also describe an association between the reduction of the subacromial space and the tear of the rotator cuff.[63] In 1984, Petersson describes the average distance of 9 to 10 mm for a normal acromio-humeral space, with a pathological threshold suspecting a lesion of the supraspinatus at 6 mm.[64] Thus, in the years that followed, many authors came to the same conclusion with a widely described association between a subacromial space less than 7 mm and a massive rupture of the rotator cuff[65][66][67][68][69] with a massive rupture of the rotator cuff described as a tear of at least two tendons, often the supraspinatus and infraspinatus.[70] Thus, Saupe et al. show in the study a strong association of the decrease of the subacromial space to less than 7 mm and a tear of the supraspinatus and infraspinatus.[65] Nové-Josserand et al. explain that a decrease of the subacromial space to less than 7 mm indicates a rupture of the infraspinatus. The reduction of the space is due to the loss of the infraspinatus lowering function with a migration of the humeral head in the space deserted by the supraspinatus. Furthermore, there is a higher rate of recurrence of rupture after surgery in a patient with a massive rupture of the cap and therefore a decreased subacromial space. Moreover, the radiological measurement showed a very good correlation whether it is compared to the scanner[71] or when comparing inter-observers.[72][73][74] All these elements therefore make it possible to use the measurement of the subacromial space in the evaluation and the therapeutic decision-making of a patient with shoulder pain. However, this remains a good indicator in the evaluation of the posterior superior cuff without giving information on the anterior cuff.[75] In connection with the reduction of subacromial space, Hamada et al. introduced in 1990 the notion of arthropathy linked to a massive rupture of the rotator cuff.[7] It declines in 5 grades progressive, each associated with radiological changes. In 2005, Walch et al. modified this classification somewhat by creating two subtypes of grade 4. However, the same year, Nové-Josserand et al. show that there is no linear progression of the Hamada classification.[76] However, it confirms that the tendinopathy of the rotator cuff is more involved in the aging of the shoulder than the osteoarthritis. A new study proposed in 2011 by Hamada et al. examines in more detail the different implications in terms of grades.[77] The greater role of a lesion involving the subscapularis from Grade 3 and a rate of recurrence of rupture after intervention more frequent from Grade 2. Thus, surgery should be considered before the subacromial space is reduced.
Ultrasound (US)
Following X-ray evaluation, advanced imaging modalities are obtained to confirm and plan treatment. Ultrasonography is an excellent cost-effective screening tool in the office but does not allow evaluation of intra-articular pathology or easy evaluation of muscle quality. Full-thickness tears extend from bursal to the articular surface, while partial-thickness tears are focal defects in the tendon that involve either the bursal or articular surface. Full-thickness appear on ultrasound as hypoechoic/anechoic defects in the tendon. Due to the fluid replacing tendon, cartilage shadow gets accentuated giving a double cortex or cartilage interface sign. Also, due to the defect, overlying peribursal fat dips down into the tendon gap, creating a sagging peribursal fat sign.[78]
Direct signs are:
- non-visualization of the supraspinatus tendon
- hypoechoic discontinuity in the tendon
Indirect signs are:
- double cortex sign
- sagging peribursal fat sign
- compressibility
- muscle atrophy
Secondary associated signs are:
- cortical irregularity of greater tuberosity
- shoulder joint effusion
- Milky way sign
- fluid along the biceps tendon
- fluid in the axillary pouch and posterior recess
Rotator Cuff Interval
The space through which the long head of the biceps passes as it leaves the glenohumeral joint is called the rotator cuff interval. The patient position is the same as for evaluation of the long head of the biceps, with the probe being placed slightly superiorly to the bicipital groove and in the axial plane (Figure). The long head of the biceps is thus visualized with the subscapularis medially and the supraspinatus laterally, while the coracohumeral and superior glenohumeral ligaments surround it.[79]
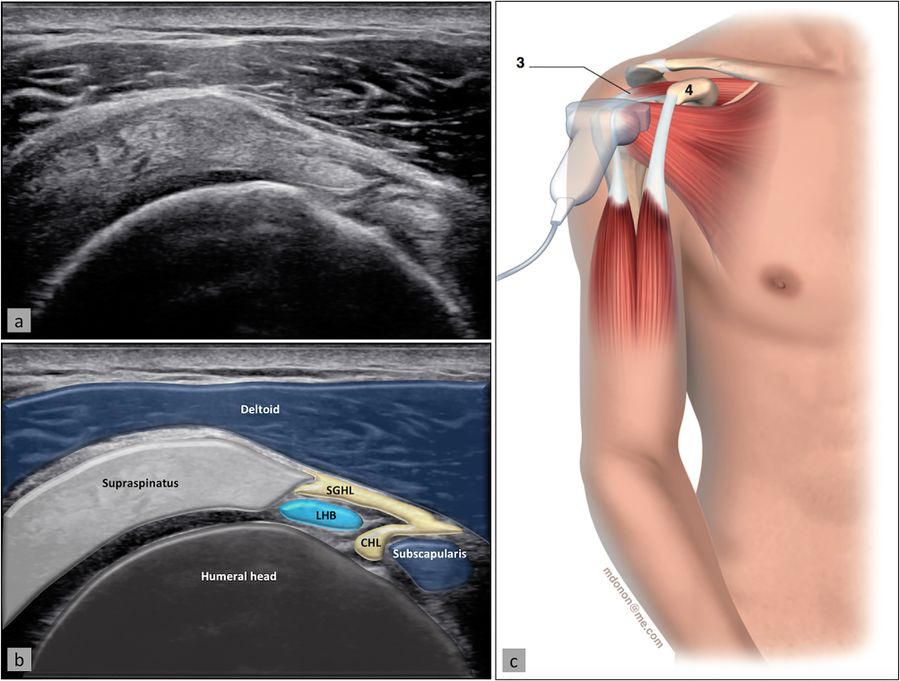
Subscapularis
Ultrasound (US) can be used as a non-invasive test to screen for subscapularis tears. However, the sensitivity of this methodology is also low.[80]
Using ultrasound imaging, the subscapularis tendon is best visualised with the shoulder in external rotation, elbow kept firmly by the thorax (Figure), drawing the tendon out from beneath the coracoid process. While looking at the tendon’s long axis, its integrity can be analyzed, as well as its relationship with surrounding structures by dynamically performing internal and external rotation of the shoulder. Examination of the short axis of the tendon (by rotating the probe by 90 degrees) reveals its multifascicular pattern, and allows complete analysis from its upper border (with the biceps tendon lying supero-laterally), where injuries often begin, to its lower border.[81]
Supraspinatus Tendon and Subacromial-Subdeltoid Bursa
The supraspinatus tendon is best visualized with the shoulder in abduction and internal rotation, by asking the patient to place the palm of their hand on their back pocket, elbow pointed backwards (Figure 8). In patients presenting with reduced range of motion (adhesive capsulitis for example), maximal internal rotation with the arm hanging by the side of the thorax can be sufficient. The long axis of the tendon is most useful for analyzing integrity of the tendon on the footprint (measuring approx. 2 cm medially to laterally), and is visualized by holding the probe in a tilted position (therefore not a true coronal plane but at an approx. 45 degree angle, following the line of the humerus).
This position also allows visualization of two other structures: the subacromial-subdeltoid bursa (and the presence of excessive liquid, see below) and the humeral head along with its articular cartilage (and possible surface defects). In the axial plane (again not truly axial but at 90 degrees to the previous plane), the leading edge of the supraspinatus can be identified laterally to the biceps tendon. Moving the probe laterally will reveal the mid-portion of the tendon, with the anterior part of the infraspinatus eventually coming into view as an anisotropic and dark image (as the fibers run in a different plane).
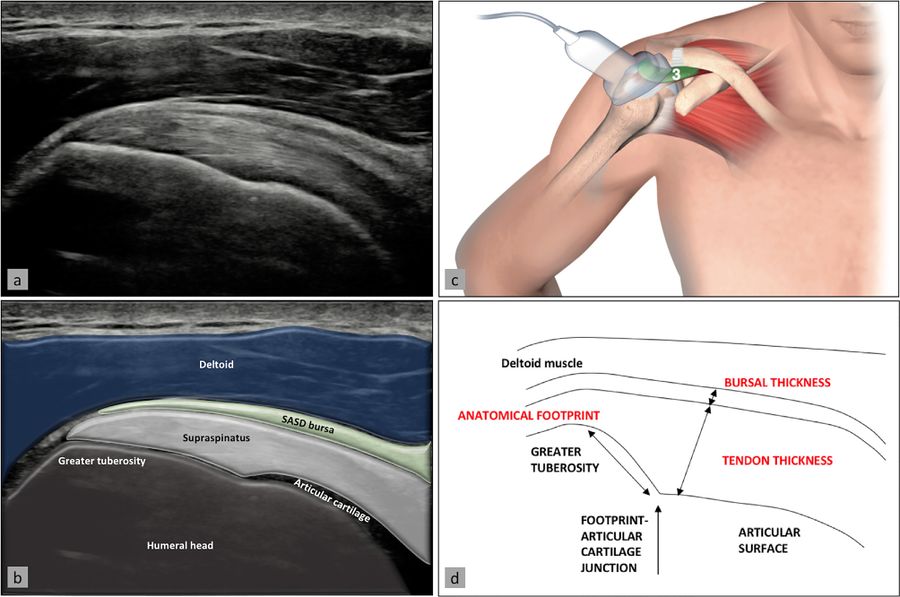
Ultrasound image (a) with superimposed anatomy (b), patient/probe position (c), and landmarks for measurement of these two structures (d). Reproduced from Plomb-Holmes et al.,[79] with permission
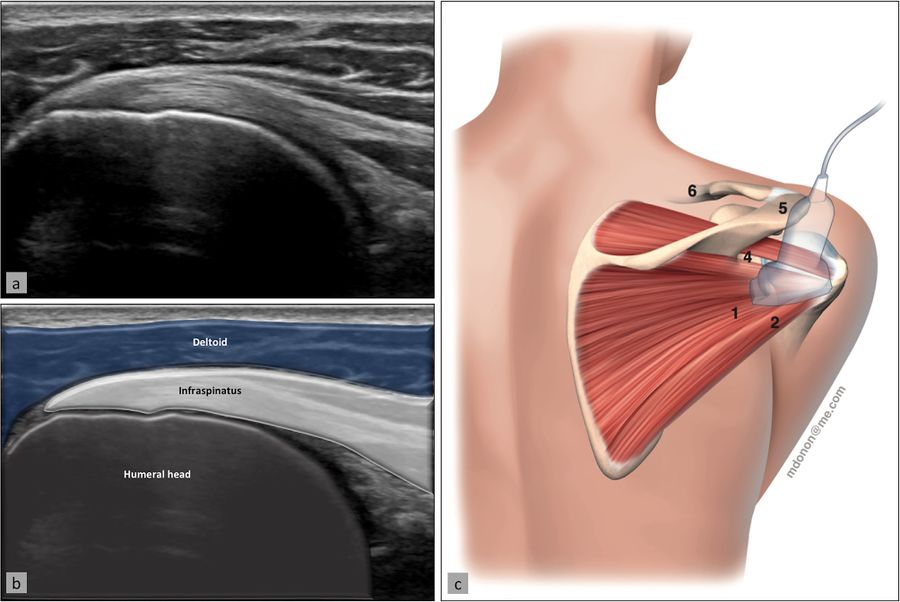
Infraspinatus and teres minor tendon, glenohumeral joint, spinoglenoid notch
The infraspinatus tendon, which inserts posteriorly to the supraspinatus tendon, is best examined in its long axis by elongating it (the patient placing his or her hand on the opposite shoulder) and placing the probe on the posterior part of the patient’s shoulder (Figure). The insertion of the tendon on the humeral head can be analyzed, as well as the musculotendinous junction by sliding the probe medially. At this point, the glenohumeral joint line and posterior labrum can be visualized in thin patients, and even more medially, the spinoglenoid notch containing the suprascapular neurovascular bundle (and the possible presence of a ganglion cyst arising from the posterior labrum which can compress the bundle) (Figure 9). The teres minor tendon can be difficult to separate from the infraspinatus tendon; it is located inferiorly and has a similar aspect, but can be distinguished by the fact that deeper to it lies bone whereas the infraspinatus lies on articular cartilage, and its insertion is primarily muscular (vs. tendinous).
Magnetic Resonance Imaging (MRI) and Computer Tomography (CT)
Magnetic resonance imaging accurately estimates tear pattern, muscle fatty infiltration and atrophy, tendon length and retraction, and is thus obtained to plan repair or reconstructive surgeries. The muscle bellies of the rotator cuff are assessed, if available, on T1-weighted axial, coronal, sagittal views with cuts sufficiently medial on the scapula to allow proper assessment regardless of retraction. Finally, computer tomography scans are used if magnetic resonance imaging is contraindicated or if joint replacement is planned, particularly in the setting of glenoid deformity. Additionally, computer tomography scan can be conducted with intra-articular contrast to assess the cuff. It should be noted that the magnetic resonance imaging and computer tomography are not reliable to analyze the acromiohumeral distance as they are performed in lying position.
Fatty Infiltration
The most important negative prognostic factor is high-grade fatty infiltration of the rotator cuff muscle bellies (grade 3 or 4 fatty infiltration) (Figure).
Fatty infiltration is irreversible even with repair and leads to reduced function of the rotator cuff musculature.[82]
If pathology of the acromioclavicular joint is suspected, a Zanca view is additionally acquired.[47][33][49]
Atrophy
The presence or absence of supraspinatus atrophy is determined using the tangent sign of Zanetti et al. (Figure 11).[83]
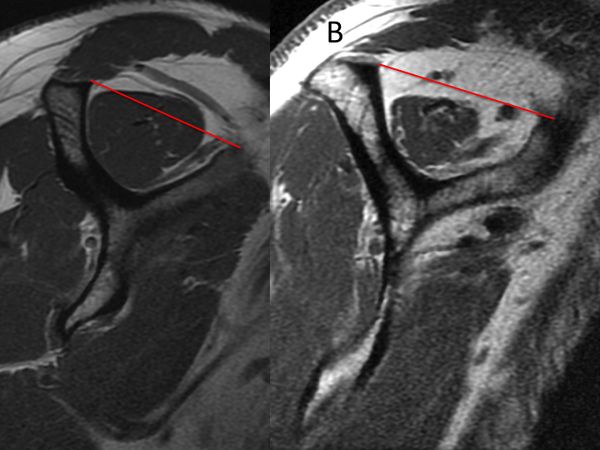
This sign is an indicator of advanced fatty infiltration and has been reported to be a predictor of whether a rotator cuff tear will be reparable.[9][45]
An inability to obtain a complete repair of the supraspinatus is associated with a positive tangent sign (30% irreparable) versus a negative tangent sign (6.3% irreparable, OR = 6,3, P =0.0102).[33]
Supraspinatus atrophy can also be determine according to Thomazeau classification.[85]
Agreement for this classification is however fair (intra-observer kappa = 0,51 and inter-observer kappa = 0.30) and its use cannot be recommended as a criteria of reparability.[86]
Conclusion
The management of a patient in the field of shoulder surgery requires a good knowledge of the anatomical and lesional representations of the standard radiography. This investigation should guide the surgeon for surgical indication or direct him towards further investigations. Although some radiological markers have demonstrated their evidence (great tuberosity displacement, subacromial space, acromial morphology and Hamada classification), others still require extensive studies and protocol standardization (acromial index, critical shoulder angle, lateral acromion angle and neck-shaft angle).
- ↑ D. Goutallier, J.M. Postel, J. Bernageau, L. Lavau, M. Voisin "Fatty muscle degeneration in cuff ruptures. Pre- and postoperative evaluation by CT scan. Clin Orthop Relat Res. 1994;304:78-83
- ↑ D. Goutallier, J.M. Postel, L. Lavau, J. Bernageau. Influence of supraspinatus and infraspinatus muscular fatty degeneration on the prognosis of rotator cuff surgical repair. Revue de Chirurgie Orthopedique et Reparatrice de l'Appareil Moteur. 1999;85:668-676
- ↑ Small KM, Rybicki FJ, Miller LR, Daniels SD, Higgins LD. MRI Before Radiography for Patients With New Shoulder Conditions. J Am Coll Radiol. 2017;14(6):778-782
- ↑ 4.0 4.1 4.2 Neer CS 2nd. Displaced proximal humeral fractures. I. Classification and evaluation. J Bone Joint Surg Am. 1970;52(6):1077-89.
- ↑ 5.0 5.1 5.2 L. Bigliani, D. Morrison, E. April "The morphology of the acromion and its relationship to rotator cuff tears." Orthop Trans, 1986, 10, 216.
- ↑ 6.0 6.1 L.U. Bigliani, J.B. Ticker, E.L. Flatow, L.J. Soslowsky, V.C. Mow "The relationship of acromial architecture to rotator cuff disease." Clin Sports Med, 1991, 10, 823-838.
- ↑ 7.0 7.1 K. Hamada, H. Fukuda, M. Mikasa, Y. Kobayashi "Roentgenographic findings in massive rotator cuff tears. A long-term observation." Clin Orthop Relat Res, 1990, 254, 92-96.
- ↑ 8.0 8.1 8.2 R.W. Nyffeler, C.M.L. Werner, A. Sukthankar, M.R. Schmid, C. Gerber "Association of a large lateral extension of the acromion with rotator cuff tears" J Bone Joint Surg Am, 2006, 88, 800-805. Cite error: Invalid
<ref>tag; name ":7" defined multiple times with different content - ↑ 9.0 9.1 9.2 B. Moor, S. Bouaicha, D. Rothenfluh, A. Sukthankar, C. Gerber "Is there an association between the individual anatomy of the scapula and the development of rotator cuff tears or osteoarthritis of the glenohumeral joint?: A radiological study of the critical shoulder angle." Bone Jt J, 2013, 95, 935-941. Cite error: Invalid
<ref>tag; name ":11" defined multiple times with different content - ↑ 10.0 10.1 J. Jeong, H.W. Jung "Optimizing intramedullary entry location on the proximal humerus based on variations of neck-shaft angle." J Shoulder Elb Surg, 2015, 24, 1386-1390.
- ↑ 11.0 11.1 M.L. Pearl "Proximal humeral anatomy in shoulder arthroplasty: Implications for prosthetic design and surgical technique." J Shoulder Elb Surg, 2005, 14, no.1 SUPPL.
- ↑ 12.0 12.1 12.2 J. Iannoti, M. Sidor "Malunions of the proximal humerus." Complex Revis Probl Shoulder Surg 1997, 245-264.
- ↑ 13.0 13.1 R. Rasmussen, I. Hvass, J. Dalsgaard, B.S. Christensen, E. Holstad "Displaced proximal humeral fractures: results of conservative treatment." Injury, 1992, 23, 41-43.
- ↑ P. Connor, E.L. Flatow "Complications of internal fixation of proximal humeral fractures." Instr Course Lect 1997, 46, 25-37.
- ↑ T.S. Park, I.Y. Choi, Y.H. Kim, M.R. Park, J.H. Shon, S.I. Kim "A new suggestion for the treatment of minimally displaced fractures of the greater tuberosity of the proximal humerus." Bull Hosp Jt Dis, 1997, 56, 171-176.
- ↑ P. Platzer, F. Kutscha-Lissberg, S. Lehr, V. Vecsei, C. Gaebler "The influence of displacement on shoulder function in patients with minimally displaced fractures of the greater tuberosity." Injury, 2005, 36, 1185-1189.
- ↑ A. De Palma, R. Cautilli, "Fractures of the upper end of the humerus" Clin Orthop, 1961, 20, 73-93.
- ↑ C. Bono, R. Renard, R. Levine "Effect of displacement of fractures of the greater tuberosity on the mechanics of the shoulder." J Bone Joint Surg Br, 2001, 83, 1056-1062.
- ↑ J. Mutch, G.Y. Laflamme, N. Hagemeister, A. Cikes, D.M. Rouleau "A new morphological classification for greater tuberosity fractures of the proximal humerus: Validation and clinical implications." Bone Joint J, 2014, 96B, 646-651.
- ↑ D.M. Rouleau, J. Mutch, G.-Y. Laflamme "Surgical Treatment of Displaced Greater Tuberosity Fractures of the Humerus" J Am Acad Orthop Surg, 2016, 24, 46-56.
- ↑ T.J. Gill, P. Waters "Valgus osteotomy of the humeral neck: a technique for the treatment of humerus varus." J Shoulder Elbow Surg, 1997, 6, 306-310.
- ↑ E. Benegas, A. Z. Filho, A.A.F. Filho, A.A.F. Neto, J.H. Negri, F.S. Prada, A.V. Zumiotti "Surgical treatment of varus malunion of the proximal humerus with valgus osteotomy." J Shoulder Elbow Surg, 2007, 16, 55-59.
- ↑ F. Hardeman, P. Bollars, M. Donnelly, J. Bellemans, S. Nijs "Predictive factors for functional outcome and failure in angular stable osteosynthesis of the proximal humerus." Injury, 2012, 43, 153-158.
- ↑ C. Voight, S. Kreienborg, O. Megatli, A.-P. Schulz, H. Lill, C. Hurschler "How does a varus deformity of the humeral head affect elevation forces and shoulder function? A biomechanical study with human shoulder specimens." J Orthop Trauma, 2011, 25, 399-405.
- ↑ F. Duparc "Malunion of the proximal humerus." Orthop Traumatol Surg Res 2013, 99, no. 1 Suppl.
- ↑ P. Boileau, G. Walch "The three-dimensional geometry of the proximal humerus. Implications for surgical technique and prosthetic design." J Bone Joint Surg Br, 1997, 79, 857-865.
- ↑ R. Hertel, U. Knothe, F. T. Ballmer "Geometry of the proximal humerus and implications for prosthetic design." J Shoulder Elbow Surg, 2002, 11, 331-338.
- ↑ E.A. Malavolta, J.H. Assuncao, R.A. Pagotto, R.L. Avelino, M.E.C. Gracetilli, C. A.M. Pereira, A.J. Jacomo, A.A.F. Neto "The rotation of the humeral head does not alter radiographic evaluation of the head-shaft angle." J Shoulder Elbow Surg, 2016, 25, 543-547.
- ↑ C. Hengg, P. Mayrhofer, S. Euler, M. Wambacher, M. Blauth, F. Kralinger "The relevance of neutral arm positioning for true ap-view X-ray to provide true projection of the humeral head shaft angle." Arch Orthop Trauma Surg, 2016, 136, 213-221.
- ↑ A. Adikrishna, H. Hong, M.F. Deslivia, B. Zhu, J. Tan, I.-H. Jeon "Head-shaft angle changes during internal and external shoulder rotations: 2-D angulation in 3-D space." Orthop Traumatol Surg Res, 2017, 103, 159-163.
- ↑ J. Tan, H.J. Lee, I. Aminata, J.M. Chun, A.L. Kekatpure, I.H. Jeon "Radiographic landmark for humeral head rotation: A new radiographic landmark for humeral fracture fixation." Injury, 2015, 46, 666-670.
- ↑ Werner CM, Conrad SJ, Meyer DC, Keller A, Hodler J, Gerber C. Intermethod agreement and interobserver correlation of radiologic acromiohumeral distance measurements. J Shoulder Elbow Surg 2008;17:237-40.
- ↑ 33.0 33.1 33.2 33.3 33.4 Sheean AJ, Hartzler RU, Denard PJ, Lädermann A, Sanders TG, Zlatkin MB, Burkhart SS. Preoperative Radiographic Risk Factors for Incomplete Arthroscopic Supraspinatus Tendon Repair in Massive Rotator Cuff Tears. Arthroscopy. 2018;34:1121-7. Cite error: Invalid
<ref>tag; name ":8" defined multiple times with different content - ↑ Hamid N, Omid R, Yamaguchi K, Steger-May K, Stobbs G, Keener JD. Relationship of radiographic acromial characteristics and rotator cuff disease: a prospective investigation of clinical, radiographic, and sonographic findings. J Shoulder Elbow Surg 2012;21:1289-98.
- ↑ Zanca P. Shoulder pain: involvement of the acromioclavicular joint. (Analysis of 1,000 cases). Am J Roentgenol Radium Ther Nucl Med. 1971;112:493-506
- ↑ Toivonen DA, Tuite MJ, Orwin JF.Acromial structure and tears of the rotator cuff. J Shoulder Elbow Surg. 1995;4(5):376-83
- ↑ Epstein RE, Schweitzer ME, Frieman BG, Fenlin JM Jr, Mitchell DG. Hooked acromion: prevalence on MR images of painful shoulders. Radiology. 1993;187(2):479-81
- ↑ Jacobson SR, Speer KP, Moor JT, Janda DH, Saddemi SR, MacDonald PB, Mallon WJ. Reliability of radiographic assessment of acromial morphology. J Shoulder Elbow Surg. 1995;4(6):449-53
- ↑ Park TS, Park DW, Kim SI, Kweon TH. Roentgenographic assessment of acromial morphology using supraspinatus outlet radiographs. Arthroscopy. 2001;17(5):496-501
- ↑ M. Aoki, S. Ishii, M. Usui "The slope of the acromion and rotator cuff impingement. Orthop Trans. 1986:10;228
- ↑ J.D. Zuckerman, F.J. Kummer, F. Cuomo, J. Simon, S. Rosenblum, N. Katz "The influence of coracoacromial arch anatomy on rotator cuff tears. J Shoulder Elbow Surg. 1992:1;4-14
- ↑ G.S. Kitay, J.P. Iannotti, G.R. Williams, T. Haygood, B.J. Kneeland, J. Berlin "Roentgenographic assessment of acromial morphologic condition in rotator cuff impingement syndrome. J Shoulder Elbow Surgery. 1995;4;441-448
- ↑ Banas MP, Miller RJ, Totterman S. Relationship between the lateral acromion angle and rotator cuff disease. J Shoulder Elbow Surgery. 1995;4(6):454-61
- ↑ Miyazaki AN, Fregoneze M, Santos PD, Da Silva LA, Martel ÉM, Debom LG, , Andrade ML, Checchia SL. Radiographic Study on the Acromion Index and Its Relationship with Rotator Cuff Tears. Rev Bras Ortop (English Ed). 2010;45:151-154
- ↑ 45.0 45.1 45.2 Engelhardt C, Farron A, Becce F, Place N, Pioletti DP, Terrier A.Effects of glenoid inclination and acromion index on humeral head translation and glenoid articular cartilage strain. J Shoulder Elbow Surg. 2017;26(1):157-164 Cite error: Invalid
<ref>tag; name ":12" defined multiple times with different content - ↑ Zumstein MA, Jost B, Hempel J, Hodler J, Gerber C. The clinical and structural long-term results of open repair of massive tears of the rotator cuff. J Bone Joint Surg Am . 2008;90(11):2423-31
- ↑ 47.0 47.1 47.2 Kircher J, Morhard M, Gavriilidis I, Magosch P, Lichtenberg S, Habermeyer P. Is there an association between a low acromion index and osteoarthritis of the shoulder? Int Orthop. 2010;34(7):1005-10 Cite error: Invalid
<ref>tag; name ":9" defined multiple times with different content - ↑ Ames JB, Horan MP, Van der Meijden OA, Leake MJ, Millett PJ. Association between acromial index and outcomes following arthroscopic repair of full-thickness rotator cuff tears. J Bone Joint Surg Am. 2012;94(20):1862-9
- ↑ 49.0 49.1 49.2 Hamid N, Omid R, Yamaguchi K, Steger-May K, Stobbs G, Keener JD. Relationship of radiographic acromial characteristics and rotator cuff disease: a prospective investigation of clinical, radiographic, and sonographic findings. J Shoulder Elbow Surg. 2012;21(10):1289-98 Cite error: Invalid
<ref>tag; name ":10" defined multiple times with different content - ↑ Hughes RE, Bryant CR, Hall JM, Wening J, Huston LJ, Kuhn JE, Carpenter JE, Blasier RB. Glenoid inclination is associated with full-thickness rotator cuff tears. Clin Orthop Relat Res. 2003;(407):86-91
- ↑ Wong AS, Gallo L, Kuhn JE, Carpenter JE, Hughes RE. The effect of glenoid inclination on superior humeral head migration. J Shoulder Elbow Surg. 2003;12(4):360-4.
- ↑ Gerber C, Snedeker JG, Baumgartner D, Viehöfer AF. Supraspinatus tendon load during abduction is dependent on the size of the critical shoulder angle: A biomechanical analysis. J Orthop Res. 2014;32(7):952-7
- ↑ Moor BK, Röthlisberger M, Müller DA, Zumstein MA, Bouaicha S, Ehlinger M, Gerber C.Age, trauma and the critical shoulder angle accurately predict supraspinatus tendon tears. Orthop Traumatol Surg Res. 2014;100(5):489-94
- ↑ Daggett M, Werner B, Collin P, Gauci MO, Chaoui J, Walch G. Correlation between glenoid inclination and critical shoulder angle: a radiographic and computed tomography study. J Shoulder Elbow Surg. 2015;24(12):1948-53
- ↑ Viehöfer AF, Snedeker JG, Baumgartner D, Gerber C. Glenohumeral joint reaction forces increase with critical shoulder angles representative of osteoarthritis-A biomechanical analysis. J Orthop Res. 2016;34(6):1047-52
- ↑ Blonna D, Giani A, Bellato E, Mattei L, Caló M, Rossi R, Castoldi F. Predominance of the critical shoulder angle in the pathogenesis of degenerative diseases of the shoulder. J Shoulder Elbow Surg. 2016 Aug;25(8):1328-36
- ↑ 57.0 57.1 Spiegl UJ, Horan MP, Smith SW, Ho CP, Millett PJ. The critical shoulder angle is associated with rotator cuff tears and shoulder osteoarthritis and is better assessed with radiographs over MRI. Knee Surg Sports Traumatol Arthrosc. 2016;24(7):2244-51
- ↑ 58.0 58.1 Cherchi L, Ciornohac JF, Godet J, Clavert P, Kempf JF. Critical shoulder angle: Measurement reproducibility and correlation with rotator cuff tendon tears. Orthop Traumatol Surg Res. 2016;102(5):559-62
- ↑ Chalmers PN, Salazar D, Steger-May K, Chamberlain AM, Yamaguchi K, Keener JD. Does the Critical Shoulder Angle Correlate With Rotator Cuff Tear Progression? Clin Orthop Relat Res. 2017;475(6):1608-1617
- ↑ Bouaicha S, Ehrmann C, Slankamenac K, Regan WD, Moor BK. Comparison of the critical shoulder angle in radiographs and computed tomography. Skeletal Radiol. 2014;43(8):1053-6
- ↑ Suter T, Gerber Popp A, Zhang Y, Zhang C, Tashjian RZ, Henninger HB. The influence of radiographic viewing perspective and demographics on the critical shoulder angle. J Shoulder Elbow Surg. 2015;24(6):e149-58
- ↑ Golding FC. The shoulder--the forgotten joint. Br J Radiol. 1962;35:149-58
- ↑ Weiner, DS, Macnab I. Superior migration of the humeral head. A radiological aid in the diagnosis of tears of the rotator cuff. J Bone Joint Surg Br. 1970;52(3):524-7
- ↑ Petersson CJ, Redlund-Johnell I.The subacromial space in normal shoulder radiographs. Acta Orthop Scand. 1984;55(1):57-8
- ↑ 65.0 65.1 Saupe N, Pfirrmann CW, Schmid MR, Jost B, Werner CM, Zanetti M. Association between rotator cuff abnormalities and reduced acromiohumeral distance. AJR Am J Roentgenol. 2006;187(2):376-82
- ↑ Nové-Josserand L, Edwards TB, O'Connor DP, Walch G. The acromiohumeral and coracohumeral intervals are abnormal in rotator cuff tears with muscular fatty degeneration. Clin Orthop Relat Res. 2005;(433):90-6
- ↑ Mayerhoefer ME, Breitenseher MJ, Wurnig C, Roposch A. Shoulder impingement: relationship of clinical symptoms and imaging criteria. Clin J Sport Med. 2009;19(2):83-9
- ↑ Nové-Josserand L, Lévigne C, Noël E, Walch G. [The acromio-humeral interval. A study of the factors influencing its height]. Rev Chir Orthop Reparatrice Appar Mot. 1996;82(5):379-85.
- ↑ Scheyerer MJ, Brunner FE, Gerber C.The acromiohumeral distance and the subacromial clearance are correlated to the glenoid version. Orthop Traumatol Surg Res. 2016;102(3):305-9
- ↑ Gerber C, Fuchs B, Hodler J. The results of repair of massive tears of the rotator cuff. J Bone Joint Surg Am. 2000;82(4):505-15
- ↑ van de Sande MAJ, Rozing PM. Proximal migration can be measured accurately on standardized anteroposterior shoulder radiographs. Clin Orthop Relat Res. 2006;443:260-5
- ↑ Werner CM, Conrad SJ, Meyer DC, Keller A, Hodler J, Gerber C. Intermethod agreement and interobserver correlation of radiologic acromiohumeral distance measurements. J Shoulder Elbow Surg. 2008;17(2):237-40
- ↑ Gruber G, Bernhardt GA, Clar H, Zacherl M, Glehr M, Wurnig C. Measurement of the acromiohumeral interval on standardized anteroposterior radiographs: a prospective study of observer variability. J Shoulder Elbow Surg. 2010;19(1):10-3
- ↑ Bernhardt GA, Glehr M, Zacherl M, Wurnig C, Gruber G. Observer variability in the assessment of the acromiohumeral interval using anteroposterior shoulder radiographs. Eur J Orthop Surg Traumatol. 2013;23(2):185-90
- ↑ Middernacht B, Winnock de Grave P, Van Maele G, Favard L, Molé D, De Wilde L. What do standard radiography and clinical examination tell about the shoulder with cuff tear arthropathy? J Orthop Surg Res. 2011;6:1
- ↑ Nové-Josserand L, Walch G, Adeleine P, Courpron P. [Effect of age on the natural history of the shoulder: a clinical and radiological study in the elderly]. Rev Chir Orthop Reparatrice Appar Mot. 2005;91(6):508-14
- ↑ Hamada K, Yamanaka K, Uchiyama Y, Mikasa T, Mikasa M. A radiographic classification of massive rotator cuff tear arthritis. Clin Orthop Relat Res. 2011 Sep;469(9):2452-60
- ↑ Moosikasuwan JB, Miller TT, Burke BJ. Rotator cuff tears: clinical, radiographic, and US findings. Radiographics. 2005;25(6):1591-607
- ↑ 79.0 79.1 79.2 79.3 Plomb-Holmes C, Clavert P, Kolo F, Tay E, Ladermann A, French Society of A. An orthopaedic surgeon's guide to ultrasound imaging of the healthy, pathological and postoperative shoulder. Orthop Traumatol Surg Res. 2018;104:S219-S232.
- ↑ Singisetti K, Hinsche A. Shoulder ultrasonography versus arthroscopy for the detection of rotator cuff tears: analysis of errors. Journal of orthopaedic surgery 2011;19:76-9.
- ↑ Plomb-Holmes C, Clavert P, Kolo F, Tay E, Ladermann A, French Society of A. An orthopaedic surgeon's guide to ultrasound imaging of the healthy, pathological and postoperative shoulder. Orthop Traumatol Surg Res. 2018 Dec;104(8S):S219-S232.27.
- ↑ Gladstone JN, Bishop JY, Lo IK, Flatow EL. Fatty infiltration and atrophy of the rotator cuff do not improve after rotator cuff repair and correlate with poor functional outcome. Am J Sports Med 2007;35:719-28.
- ↑ Zanetti M, Gerber C, Hodler J. Quantitative assessment of the muscles of the rotator cuff with magnetic resonance imaging. Investigative radiology 1998;33:163-70
- ↑ Lädermann A, Collin P, Athwal GS, Scheibel M, Zumstein MA, Nourissat G. Current concepts in the primary management of irreparable posterosuperior rotator cuff tears without arthritis. EFORT Open Rev. 2018;3:200-9
- ↑ Thomazeau H, Rolland Y, Lucas C, Duval JM, Langlais F. Atrophy of the supraspinatus belly. Assessment by MRI in 55 patients with rotator cuff pathology. Acta Orthop Scand 1996;67:264-8
- ↑ Niglis L, Dosch JC. Intra- and inter-observer agreement in MRI assessment of rotator cuff healing using the Sugaya, Goutallier, Warner and Thomazeau classifications 10 years after surgery. “s.l.” and “s.n.”: Université de Strasbourg; 2015